Is N2O linear or bent?
2 Answers. Nitrous oxide is linear.
Why is N2O linear?
In the diagram given above, the type of bonding in nitrous oxide is covalent bonding. Due to the presence of sigma bonds and absence of lone pairs on the central atom, the stable structure of nitrous acid is linear. It is also known as sulphur dioxide.
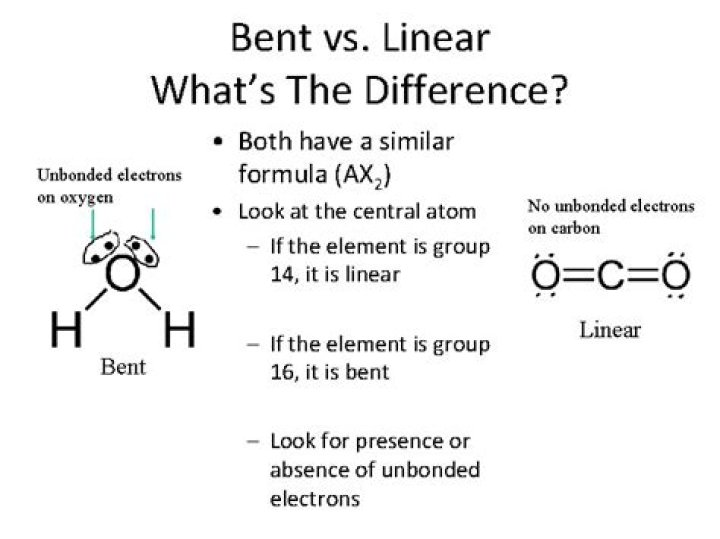
How do you know if its bent or linear?
1st remember the names: The names can be determined by the shape and angle of the molecule. Linear = is just a line of atoms with a 180° angle. Notice that it’s 2 or 3 atoms total. Bent = Linear but bent due to the Lone Pairs that it contains, the more Lone Pairs the greater the bent and the smaller the degree.
N2O is a neutral molecule, nitrous oxide, it is isoelectronic with CO2. The intermolecular force which polar molecules take part in are dipole-dipole forces.
What is the bond angle of N2O?
The 18 electrons must be distributed in such a way that all the atoms get 8 electron each. The lewis structure for the molecule is: From the above structure, it is clear that N2O has linear structure. Thus, the bond angle in is 180°.
The molecular shape of hydrogen sulfide is bent. The central atom sulfur is bonded to two hydrogen atoms.
How do you find the linear structure?
The number of bonded pairs is equal to the number of substrates attached to the central element of the binary compound and represents the number of ‘Bonded Pairs’ of electrons. #BPrs = 2. => AX2 => Linear Geometry X – A – X for the BeCl2 structure. That is, Cl – Be – Cl is a linear molecule.
Is H2S bent?
Thus, we can say that the molecular geometry of H2S is bent. Moving to the difference between electron geometry and molecular geometry. Basically, molecular geometry only takes into account the atoms of the molecules while determining the shape. Whereas electron geometry considers all the electrons present.
The given molecule is,
The nitrogen has ‘5’ valence electrons and oxygen has ‘6’ valence electron. According to electron-dot structure, there are 8 number of bonding electrons and 8 number of non-bonding electrons. In the structure of , one N molecule act as a central atom and is bonded to another N and O atom.
Does N2O have linear structure?
is N2O molecule linear in structure? yes. N2O molecule has a linear shape.
What type of bond does N2O have?
The nitrous oxide molecule contains a total of 2 bond(s) There are 2 non-H bond(s), 2 multiple bond(s), 1 double bond(s) and 1 triple bond(s). The 2D chemical structure image of nitrous oxide is also called skeletal formula, which is the standard notation for organic molecules.



