Is methanol a polar or nonpolar molecule?
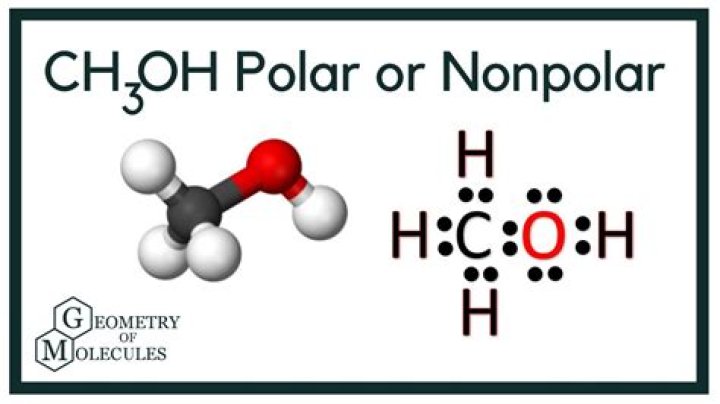
Any molecule with lone pairs of electrons around the central atom is polar. Methanol is polar. This is not a symmetric molecule. The −OH side is different from the other 3 −H sides.
Is CH3OH polar or nonpolar molecule?
CH3OH is a polar molecule as the dipole-dipole moment is not canceled due to its asymmetric shape.
Is Benzhydrol polar or nonpolar?
So, it is completely a non-polar compound. Benzhydrol contains an alcohol functional group. It is attached to the two phenyl rings. So, it has the polar character due to the alcoholic functional group but the non-polar part is also there as two benzene rings are present.
CH3F is a polar molecule, even though the tetrahedral geometry often leads to nonpolar molecules.
What bond type is methanol?
The type of bonding found in methanol is hydrogen bonding. Methanol is polar, which exhibits dipole interaction.
The molecular geometry around oxygen in methanol is bent. Oxygen is more electronegative than carbon or hydrogen, so the electron density is skewed towards oxygen. Therefore, there is a net dipole with the negative end pointing through oxygen, and methanol is polar.
Why is methanol polar?
Methanol is a polar molecule:
The alcohol (-OH) group dominates the molecule making it definitely polar. The electrostatic potential clearly shows that the oxygen is partially negative whereas the carbon and hydrogens are partially positive.
What makes a molecule polar?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A dipole is any molecule with a positive end and a negative end, resulting from unequal distribution of electron density throughout the molecule.
Hexane is a non-polar solvent with a boiling point of 68°C, and is therefore the solvent of choice for oil extraction from rice bran to yield rice bran oil (RBO).
Is Diphenylmethanol polar?
The answer is of course an hydroxyl substituent, a strongly POLAR functional group; i.e. (H5C6)2CH(OH) versus O=C(C6H5)2 .
Is Fluoromethane methyl fluoride polar or nonpolar?
CH3F is a polar molecule due to the presence of higher electronegative Fluorine atom and gains a partial negative charge and other atoms gain partial positive charge and make the molecule polar.
Is H2 polar?
Covalent molecules made of only one type of atom, like hydrogen gas (H2), are nonpolar because the hydrogen atoms share their electrons equally. 3-6 for examples of polar and nonpolar molecules.
BrF5 or Bromine Pentafluoride is a polar molecule as the molecular geometry of BrF5 falls out to be square pyramidal with an asymmetric charge distribution concentrating on the central atom. Therefore this molecule is ought to be polar.
Is methanol a molecule?
Methanol, also known as methyl alcohol, carbinol, wood alcohol, wood naphtha or wood spirits, is a chemical compound with chemical formula CH3OH (often abbreviated MeOH).
Is methanol more polar than ethanol?
Yes, like Mohammed said, methanol is more polar than ethanol which is more polar than butanol. As for solvent fractionation of extracts, there are many ways to do it.
How many polar bonds are in methanol?
c. The oxygen in methanol is involved in two polar bonds, so we will focus on the geometry about this atom. The geometry about the oxygen (O) in methanol is tetrahedral (four groups of electrons).
Methanol has a higher boiling point than methane because it has stronger intermolecular forces (IMFs), which are attractions between individual molecules. Methane is thus a polar molecule. When hydrogen is bonded to one of the three most electronegative elements, F, O or N, the molecule exhibits hydrogen bonding.
Is methanol or water more polar?
Methanol 100% is a good organic solvent to extract most of the compounds but it is not that polar compared to water.
Is methanol hydrophobic or hydrophilic?
alcohols. …is referred to as a hydrophilic (“water-loving”) group, because it forms hydrogen bonds with water and enhances the solubility of an alcohol in water. Methanol, ethanol, n-propyl alcohol, isopropyl alcohol, and t-butyl alcohol are all miscible with water.



