Is methane a polar or nonpolar molecule?
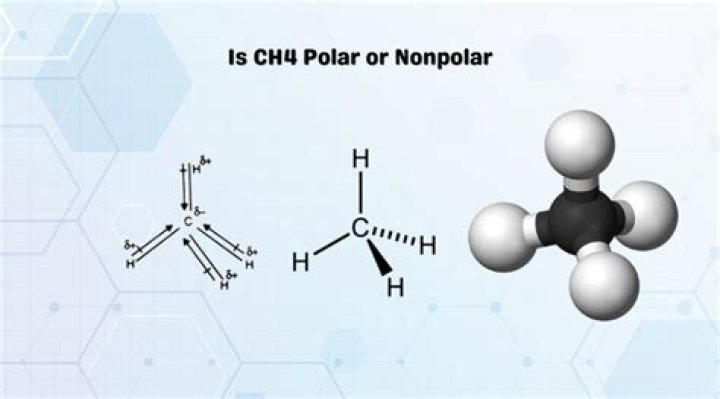
As we have seen, the C-H bonds in methane are polar. However, a molecule of methane is non-polar. Specifically, the dipole moment of methane is zero. A dipole moment of zero means that the “center of negative charge” in the molecule corresponds to the “center of positive charge”.
Why methane is a nonpolar molecule?
When a molecule is Polar they have two different centres of positive and negative charges. Methane Does not have the charges present at the end because of finely distributed electrons and this distribution which is symmetrical cancel out each other. Hence it is non-polar.
Is methane CH4 polar or nonpolar?
All the outer atoms are the same – the same dipoles, and that the dipole moments are in the same direction – towards the carbon atom, the overall molecule becomes non-polar. Therefore, methane has non-polar bonds, and is non-polar overall.
2 Answers By Expert Tutors. Methane contains nonpolar covalent bonds. This is because there is a very small difference in the electronegativity of Carbon and Hydrogen. Thus they are going to share their electrons fairly equally.
Is methane a polar covalent compound or nonpolar covalent compound?
Methane is said to be non-polar because the shared pair of electrons is equally distributed between all bonding atoms of methane.
Methane (CH4) is a non-polar hydrocarbon compound composed out of a single carbon atom and 4 hydrogen atoms. Methane is non-polar as the difference in electronegativities between carbon and hydrogen is not great enough to form a polarized chemical bond.
Is BCl3 polar or nonpolar?
BCl3 B C l 3 has trigonal planar geometry. There are three polar B-Cl bonds in this compound but due to the symmetry in its structure, all the bond dipoles of polar bonds cancel each other resulting in a zero resultant dipole moment of the molecule. So, BCl3 B C l 3 is polar.
Why is methane a covalent molecule?
Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram). There are four bonds from a central carbon (C) linking or bonding it to four hydrogen atoms (H). The methane molecule is this group of 5 atoms connected as such.
Gases such as methane are actually hydrophobic, yet the hydrophobic guests are essential in stabilizing the overall structure.
What type of bond does methane have?
Methane has four covalent bonds between carbon (C) and hydrogen (H).
What’s the structure of methane CH4 is it polar or non-polar Brainly?
Methane(CH4) is a non-polar hydrocarbon compound composed out of a single carbon atom and 4 hydrogen atoms.
Why is methane nonpolar but chloromethane is polar?
Methane isn’t non-polar because of its tetrahedral shape: it is non-polar because all the bonds are the same. Dichloromethane is polar because it has different polarity bonds and its shape cannot arrange those bond dipoles to cancel out.



