Is HOCl polar or nonpolar?
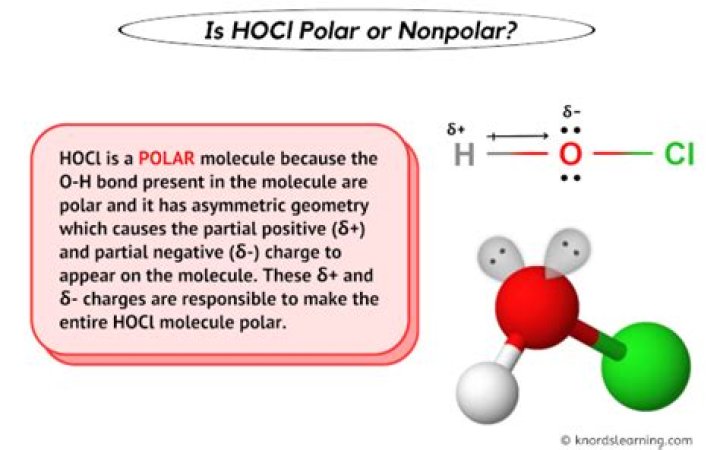
Hypochlorous acid is HOCl. Here Oxygen atom is sp3 hybridised. Hence, it has bent shape around oxygen owing to presence of two lone pairs. This causes net Dipole moment (0.37 D) and hence it is a polar molecule.
Is HOCl polar or nonpolar?
The molecule has bent shape and there are two lone pair of electrons on O atom. Therefore, overall molecule is non-polar in nature.
Does HOCl have polar bonds?
Cocoa solids are a mixture of both slightly polar and non-polar compounds and sucrose (sugar) that is extremely polar (huge dipole moment).
Is HClO a polar covalent bond?
Consider the hydrogen chloride (HCl) molecule. Each atom in HCl requires one more electron to form an inert gas electron configuration. Consequently, the bonding electrons in hydrogen chloride are shared unequally in a polar covalent bond.
Why is HCLO polar?
Hypochlorous acid is HOCl. Here Oxygen atom is sp3 hybridised. Hence, it has bent shape around oxygen owing to presence of two lone pairs. This causes net Dipole moment (0.37 D) and hence it is a polar molecule.
What shape is HOCl?
HOCl molecule is angular or ‘V’-shaped.
Is the molecule HOCl polar or nonpolar use a Lewis dot structure to explain your thinking?
We have hydrogen which has one valence electron, It is bonded to oxygen, oxygen has six valence electrons, two of them are pairs and two of them are lone electrons. And then we have chlorine Which has seven valence electrons. So two polar bonds and it’s bent. So this is definitely going to be a polar molecule.
Is HOCl a hydrogen bond?
鈻 The Cl atom of HOCl molecule can act as a hydrogen-bond or halogen-bond acceptor, or as a halogen-bond donor. 鈻 The HOCl molecule can dimerize to form six different complexes but not at room temperature.
Does HOCl have a dipole moment?
The dipole moment of HOCl in vOH=4 – ScienceDirect.
Is vegetable oil polar or nonpolar?
Vegetable oil is a non-polar molecule. These two substances do not mix together, they are imiscible (they will not mix together). That’s why you see the blobs of water bobbing around in the oil. Food coloring is a polar molecule so it WILL mix with the water.
Is theobromine a polar molecule?
The values of the dipole moment tells us that this molecules are polar with Theobromine being more polar than Theophylline.
Is chocolate polar or non polar?
It has two very different components: the small, polar flavor particles that give chocolate its taste and the white, nonpolar lipid known as cocoa butter that gives chocolate its structure.
What substances are polar?
Polar molecules occur when two atoms do not share electrons equally in a covalent bond.
Examples of polar molecules include:
Water – H2O.Ammonia – NH. Sulfur dioxide – SO. Hydrogen sulfide – H2S.Ethanol – C2H6O.
What are nonpolar covalent bonds?
Non-polar bonds are also a type of covalent bond. Unlike polar bonds, non-polar bonds share electrons equally. A bond between two atoms or more atoms is non-polar if the atoms have the same electronegativity or a difference in electronegativities that is less than 0.4.
Is Al Al a covalent bond?
Ernest Z. Aluminum and chlorine form covalent bonds, not ionic bonds.
What is the Lewis structure for HCLO?
The Lewis structure of hypochlorous acid has oxygen (O) with single bonds between hydrogen and chlorine. In the Lewis structure, we see that hypochlorous acid has 14 valence electrons. Four are used as bonding electrons, and the remaining 10 are nonbonding electrons on oxygen and chlorine.
How many valence electrons are in HOCl?
Total number of electrons of the valance shells of HOCl
Chlorine has 7 valence electrons in its valence shell. Now we know how many electrons are includes in valence shells of hydrogen, oxygen and chlorine atoms.



