Is H3O+ a planar molecule?
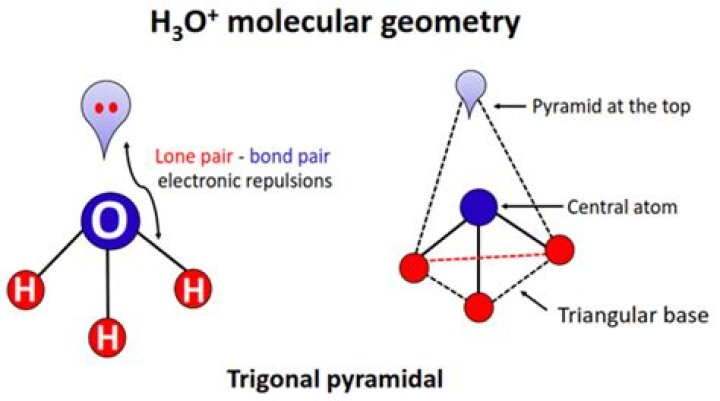
H3O + is pyramidal in shape.
Is H3O+ a planar molecule?
Note: If there would not have been any lone pair, the structure of the hydronium ion would be trigonal planar. The molecules or ions having three bond pairs of electrons and no lone pair have trigonal planar structures.
Does H3O+ have Tetrahedral geometry?
The molecular shape of H3O+ is a trigonal pyramid and electronic geometry is tetrahedral. What is this? (A= central atom, X= bonded atom, E= lone pair on A). So according to the VSEPR chart, H3O+ has trigonal pyramid as its molecular shape and tetrahedral as its electron geometry.
Is H3O trigonal?
H3O+ is has an electron arrangement tetrahedral because there are 4 regions of electron density. However, the shape of H3O+ is trigonal planar because there are 3 bonds and 1 lone pair in H3O+.
Is H3O polar or nonpolar?
H3O is the abbreviation for the hydronium ion which is considered to be polar.
What type of bond is H3O?
When water molecule combines with H+ ion by the donation of lone pair electron from O-atom (in H2O) to H+ ion results in the formation of Hydronium ion ,H3O+. Coordinate Covalent bond is a type of covalent bond that forms when one atom donates a pair of electron(Nucleophile) to another atom (Electrophile).
What is the molecular shape of ClO3?
The shape of ClO3- ion is trigonal pyramidal. According to VSEPR (Valence shell electron pair) theory.
What is the hybridisation of H3O+?
The hybridization in the central atom of H30+ is sp3 and geometry is trigonal pyramidal. Hybridization depend upon the number of Bond Pairs and Lone Pairs on the given atom. Since there are 4 regions of electron density surrounding the central O atom (3 Hydrogens and 1 lone pair of electrons).
What is the bond angle of H3O+?
The bond angle of H3O+ predicted using VSEPR (valence shell electron pair) theory would be 107o. The formation of a H3O+ ion is essentially a H+ ion attaching itself to a H2O molecule via dative bond formation.
Is H3O+ an acid or base?
When water acts as a base, it becomes H3O+, which is an acid and is called the conjugate acid of water.
How many sigma bonds does H3O+ have?
Hydronium ion has a central oxygen atom to which three hydrogen atoms are bonded. These bonds are all single bonds so there are a total of three



