Is h30 polar or nonpolar?
H3O is the abbreviation for the hydronium ion which is considered to be polar.
What is the molecular shape of H3O+?
The molecular shape of H3O+ is a trigonal pyramid and electronic geometry is tetrahedral.
Is H3O+ trigonal planar?
H3O+ is has an electron arrangement tetrahedral because there are 4 regions of electron density. However, the shape of H3O+ is trigonal planar because there are 3 bonds and 1 lone pair in H3O+.
When water acts as a base, it becomes H3O+, which is an acid and is called the conjugate acid of water.
Is h20 polar?
Water (H2O), like hydrogen fluoride (HF), is a polar covalent molecule. When you look at a diagram of water (see Fig. 3-2), you can see that the two hydrogen atoms are not evenly distributed around the oxygen atom.
Unequal Sharing
Electrons in the bonds between identical atoms (H-H) are shared uniformly, so the electrons spend equal amounts of time around each atomic center. These covalent bonds are non-polar.
Is I2 polar?
We can conclude from all the above-mentioned facts that I2 is non-polar in nature. The reason for this lies in its property of equal sharing of electrons between two iodine atoms to gain stability. Also due to the same electronegativity and linear structure, there is zero net dipole moment making I2 non-polar.
Is h20 a nonpolar molecule?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule.
HCl (hydrochloric acid) is a polar molecule because the chlorine is more electronegative than hydrogen due to which it attracts the bonded electron pair slightly nearer to it and gains a partial negative charge and hydrogen gains a partial positive charge. The dipole moment of HCl turns out to be 1.03 D.
Is NaCl polar?
Sodium Chloride (NaCl) which is an ionic compound acts as a polar molecule. Usually, the large difference in electronegativities in sodium and chlorine makes their bond polar.
Which molecule is nonpolar?
Nonpolar Molecule Examples
Examples of homonuclear nonpolar molecules are oxygen (O2), nitrogen (N2), and ozone (O3). Other nonpolar molecules include carbon dioxide (CO2) and the organic molecules methane (CH4), toluene, and gasoline. Most carbon compounds are nonpolar.
How do you tell if a molecule is polar?
If the arrangement is symmetrical and the arrows are of equal length, the molecule is nonpolar.If the arrows are of different lengths, and if they do not balance each other, the molecule is polar.If the arrangement is asymmetrical, the molecule is polar.
Any of the homonuclear diatomic elements: H2, N2, O2, Cl2 (These are truly nonpolar molecules.) Carbon dioxide – CO. Benzene – C6H. Carbon tetrachloride – CCl.
What are examples of polarity?
Examples of Polar Molecules
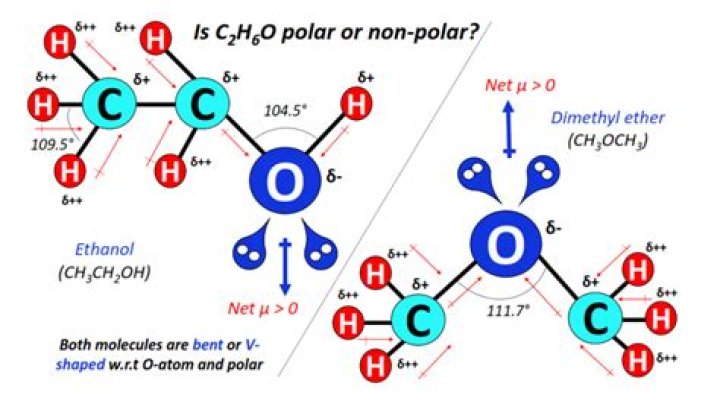
Water (H2O) is a polar molecule. Ethanol is polar because the oxygen atoms attract electrons because of their higher electronegativity than other atoms in the molecule. Ammonia (NH3) is polar.Sulfur dioxide (SO2) is polar.Hydrogen sulfide (H2S) is polar.
What makes a molecule polar?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A dipole is any molecule with a positive end and a negative end, resulting from unequal distribution of electron density throughout the molecule.
What is polar and non-polar?
When things are different at each end, we call them polar. Some molecules have positive and negative ends too, and when they do, we call them polar. If they don’t, we call them non-polar. Things that are polar can attract and repel each other (opposite charges attract, alike charges repel).



