Is H2CO3 a polar molecule?
Ernest Z. Yes, H2CO3 is a polar molecule.
How do you know if it’s a polar molecule?
If there are no lone pairs on the central atom, and if all the bonds to the central atom are the same, the molecule is nonpolar. If the central atom has at least one polar bond and if the groups bonded to the central atom are not all identical, the molecule is probably polar.
Which is a polar molecule?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A diatomic molecule that consists of a polar covalent bond, such as HF, is a polar molecule. A molecule with two poles is called a dipole (see Figure below ) . Hydrogen fluoride is a dipole.
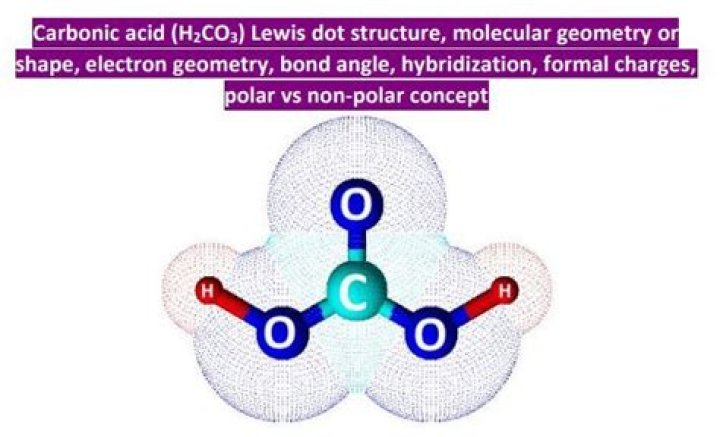
The geometrical shape of the H2CO3 molecule is trigonal planar. The Carbon and 3 Oxygen atoms lie in the same plane.
Which compound has non-polar molecules?
Nonpolar Molecule Examples
Examples of homonuclear nonpolar molecules are oxygen (O2), nitrogen (N2), and ozone (O3). Other nonpolar molecules include carbon dioxide (CO2) and the organic molecules methane (CH4), toluene, and gasoline. Most carbon compounds are nonpolar. A notable exception is carbon monoxide, CO.
All symmetrical molecules are non-polar and all asymmetrical molecules are polar.
Is SiO2 polar or nonpolar?
And that the Lewis structure of Silicon dioxide does not have any lone pairs of electrons. So there is no shape distortion, making it easy to cancel out the dipole moment produced by the Si-O bond. As a result of all of these factors, SiO2 is a non-polar molecule.
Which is the most polar molecule?
Water is the most polar molecule because a bond between oxygen and hydrogen has the most difference out of the atoms listed. Although the oxygen has two hydrogens bonded, this does not decrease the electronegativity of oxygen, but oxygen unfairly shares sets of electrons from both hydrogens, making it more polar still.
Water (H2O) is an example of a polar molecule since it has a slight positive charge on one side and a slight negative charge on the other. The dipoles do not cancel out, resulting in a net dipole. Due to the polar nature of the water molecule itself, other polar molecules are generally able to dissolve in water.
What is an example of a polar bond?
In polar covalent bonds, one atom has a stronger pull than the other atom and attracts electrons. A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms.
How is H2CO3 polar?
Out of these $ 24 $ valence electrons, $ 12 $ valence electrons were involved in bonding electrons, and remaining electrons exist as lone pairs on three oxygen atoms in carbonic acid. Thus, there is a dipole moment in the carbonic acid molecule. The presence of a dipole moment makes the molecule a polar molecule.
Is H2CO3 ionic or covalent?
Carbonic acid is the only one shown that contains six covalent bonds, making it the correct answer.
We know, when hydrogen made a covalent bond, there are no lone pairs on hydrogen atom because hydrogen cannot keep more than two electrons in its valence shell. In the lewis structure of H2CO3, carbon atom has made four bonds.



