Is H2CO a dipole?
H2CO is a polar molecule and will have both dipole-dipole forces and London dispersion forces while CH3CH3 is a non-polar molecule and will only have London dispersions forces.
Is CH2O dipole or nonpolar?
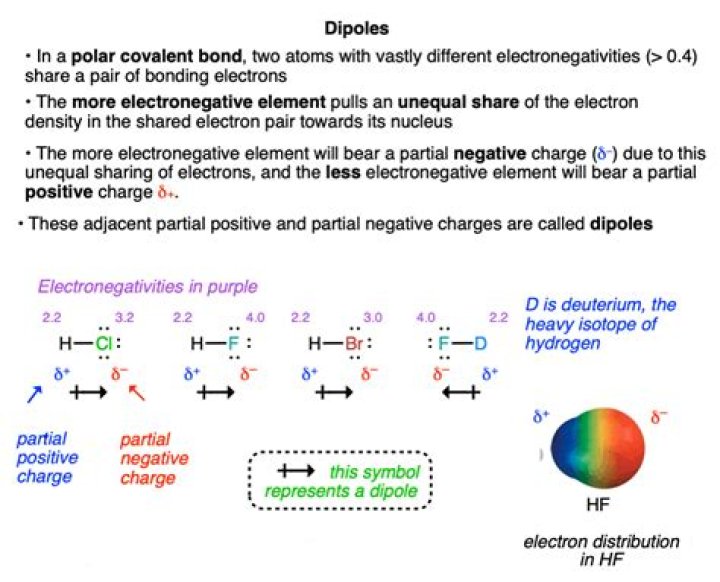
So, is CH2O polar or nonpolar? CH2O is polar in nature because of the higher electronegativity of oxygen(3.44) atom. The oxygen atom gains partial negative charge leaving behind partial positive charge on carbon and hydrogen atoms. Due to this charge imbalance, the molecule turns out to be polar.
Is h2oc polar?
H2 CO is polar because the shared electrons between the carbon atom and the oxygen atom are not shared equally between the two.
It is an organic compound with the molecular formula of H2CO and is classified as an aldehyde. Aldehydes are chemicals having the functional group -HCO- in their molecules and formaldehyde is the lowest member of this group with a single carbon atom.
What is the bond order of H2CO?
So that’s the Lewis structure for H2CO. To determine the bond order of a diatomic molecule such as H 2, CO or HCl, you simply look at the kind of bond involved and that is your answer. There is a double bond between the two oxygen atoms; therefore, the bond order of the molecule is 2.
Thus H2CO is an almost symmetric top and its rotational spectrum should resemble that of a symmetric top. However, each symmetric top transition will be asymmetry-split into several resolvable transitions.
Is CH2O a molecular dipole?
Due to their different three-dimensional structures, some molecules with polar bonds have a net dipole moment (HCl, CH2O, NH3, and CHCl3), indicated in blue, whereas others do not because the bond dipole moments cancel (BCl3, CCl4, PF5, and SF6).
What is the dipole moment of CH2O?
CH2O is soluble in water and acetone. Solubility in water is 400 g/L. Its vapor pressure is greater than 1. The dipole moment of methanal is 2.330 D.
CH2O and CH3OH are polar, so their strongest IMF are dipole – dipole; however, CH3OH can hydrogen bond while CH2O cannot so its dipole – dipole forces should be stronger.
Is CH3Br polar or nonpolar?
The CH3Br molecule is polar. Both CBr4 and CH3Br have four regions of electrons around the central carbon atom. These are all bonding electron regions (clouds) so the shape of both molecules is tetrahedral. The C-Br bond is polar due to the difference in electronegativity between C and Br.
Is H20 a electronegativity?
Polarity of a Water Molecule
Water (H2O) is polar because of the bent shape of the molecule. The electronegativity value of hydrogen is 2.1, while the electronegativity of oxygen is 3.5. The smaller the difference between electronegativity values, the more likely atoms will form a covalent bond.



