Is density of water constant?
Density is a characteristic property of water because the density of any sample of water (at the same temperature) is always the same. The density is 1 g/cm3.
Is density a constant?
The density is constant throughout, and the density of any sample of the substance is the same as its average density. If the density of a substance were not constant, the substance is said to be a heterogeneous substance.
Is the density of water constant at different temperatures?
The water density varies for different temperature. The density (in kg/m3) of water for different temperature scale (ranging from 100 °C to -30 °C) is given in the table below.
The density of water can also be affected by temperature. When the same amount of water is heated or cooled, its density changes. When the water is heated, it expands, increasing in volume. The warmer the water, the more space it takes up, and the lower its density.
Is density constant for gas?
Since the chamber is sealed and its volume is constant, the density of the reference gas remains constant. As temperature changes, the gas pressure within the reference chamber changes in order to maintain this constant density.
In practical terms, density is the weight of a substance for a specific volume. The density of water is roughly 1 gram per milliliter but, this changes with temperature or if there are substances dissolved in it. Ice is less dense than liquid water which is why your ice cubes float in your glass.
What density floats on water?
In the case of water, an object with a density less than 1 g/cm3 will float. The closer its density is to 1 g/cm3, the more of it will sit below the water level. An object with a density of 0.5 g/cm3 will sit half in and half out of the water.
How do you find the density of water?
Just like a solid, the density of a liquid equals the mass of the liquid divided by its volume; D = m/v. The density of water is 1 gram per cubic centimeter.
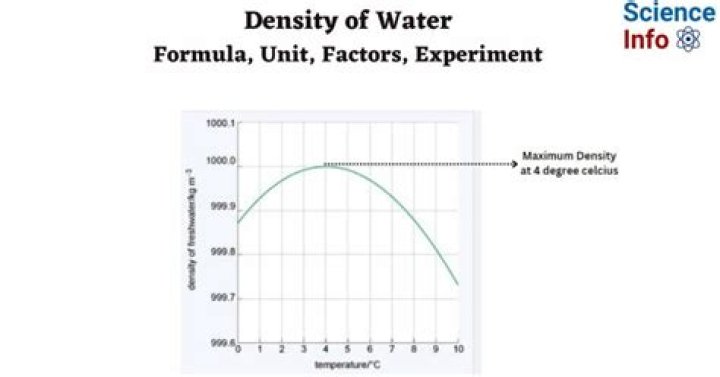
It is well known today that water has its maximum density at a temperature of about 14°C or 39°F.
Why is water densest at 4 degrees?
As the temperature of warm water decreases, the water molecules slow down and the density increases. At 4 °C, the clusters start forming. The molecules are still slowing down and coming closer together, but the formation of clusters makes the molecules be further apart. Thus, the density of water is a maximum at 4 °C.
Why density of water is more than ice?
The “stuff” (molecules) in water is more tightly packed than in ice, so water has greater density than ice. As water freezes it expands. So, ice has more volume (it takes up more space, but has less density) than water.
Is water more dense as a solid or liquid?
Solid water, or ice, is less dense than liquid water. Ice is less dense than water because the orientation of hydrogen bonds causes molecules to push farther apart, which lowers the density.
It’s All About Hydrogen Bonds
When water freezes into ice, it crystallizes into a rigid lattice that increases the space between molecules, with each molecule hydrogen bonded to 4 other molecules. “Why Is Water More Dense Than Ice?” ThoughtCo, Aug. 27, 2020,
Which phase of water is densest?
[Liquid water is densest, water vapor is least dense.] 2.



