Is co3 2 trigonal planar?
3 that the molecular geometry of CO 3 2− is trigonal planar with bond angles of 120°.
Does CO3 2 have a trigonal planar shape?
n stands for the number of bonds attached to the central atom inside the molecule, As per the VSEPR chart with AXnEx notations, we can predict our molecular shape. The shape of AX3 notation as in CO32- ion is trigonal planar with a bond angle of about 120 degrees.
What is trigonal planar examples?
Examples of Trigonal Planar Compounds
Trigonal planar geometries are also possible if the central atom of a compound shares double bonds with the other atoms. Even if the atom has a double bond, it still counts as one group. Examples include formaldehyde (CH2O), sulfur trioxide (SO3), and phosgene (COCl2).
In essence, nitrate has 3 electron domains with zero lone pairs. Thus, NO3- molecular geometry is trigonal planar and is slightly bent. The bond angle is 120°.
Why does CO3 2 have a trigonal planar shape?
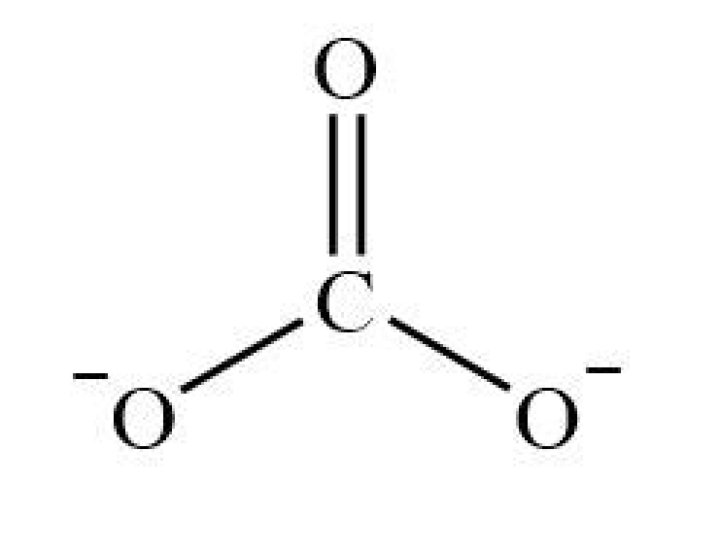
Answer: The shape of CO3^2- is trigonal planar. It has the carbon in the middle with three oxygens bonded to it. One C-O bond is a double bond, while the other two C-O bonds are single bonds. The oxygens with the single bonds have three lone pairs each and the oxygen with the double bond has two lone pairs.
Carbonate Ion:
The carbon and oxygen are bonded through a double bond which counts as “one electron pair” and two single bonded oxygens. Hence the molecule has three electron pairs and is trigonal planar geometry.
Is CO32 molecular geometry?
3 that the molecular geometry of CO 3 2− is trigonal planar with bond angles of 120°.
Is CO3 2 a polar or nonpolar molecule?
As shown above, the CO32- ion has polar bonds. However, these bonds and their constituent negative charge are distributed evenly due to their symmetric shape. This results in a zero net dipole moment. Therefore, the CO32- ion is non-polar in nature.
What is the difference between Trigonal Planar and Trigonal Pyramidal? In trigonal planar, there are no lone pair electrons in the central atom. But in trigonal pyramidal there is one lone pair at the central atom. In trigonal planar, all the atoms are in one plane but, in trigonal pyramidal they are not in one plane.
What does trigonal pyramidal look like?
Trigonal pyramidal is a molecular shape that results when there are three bonds and one lone pair on the central atom in the molecule. Molecules with an tetrahedral electron pair geometries have sp3 hybridization at the central atom. Ammonia (NH3) is a trigonal pyramidal molecule.
What is the shape of carbonate?
Carbonate ion, CO32- is trigonal planar in shape with a O-C-O bond angle of 120o because of three groups of bonding electrons and no lone pairs of electrons.
Is NO2 trigonal planar?
In this example, NO2, the Lewis diagram shows nitrogen at the center with one lone electron. Hence the molecule has three electron pairs and is trigonal planar for electron pair geometry.
3. Geometrical or molecular shape: We know the molecular shape of SiO2 is linear which is highly symmetrical and the lewis structure of Silicon dioxide doesn’t have any lone pair of electrons so there is no distortion of shape happen which made it easy to cancel out the dipole moment generated along with the Si-O bond.
How many double bonds are in co32?
Each carbon oxygen bond can be thought of as 1.333 bonds. the average of a double bond and 2 single bonds. 4 bonds/3 structures.
How many bonds are in co32?
There are three σ bonds and π bond around carbon atom in the Lewis structure of CO32- ion.



