Is CH3OH a hydrogen bond?
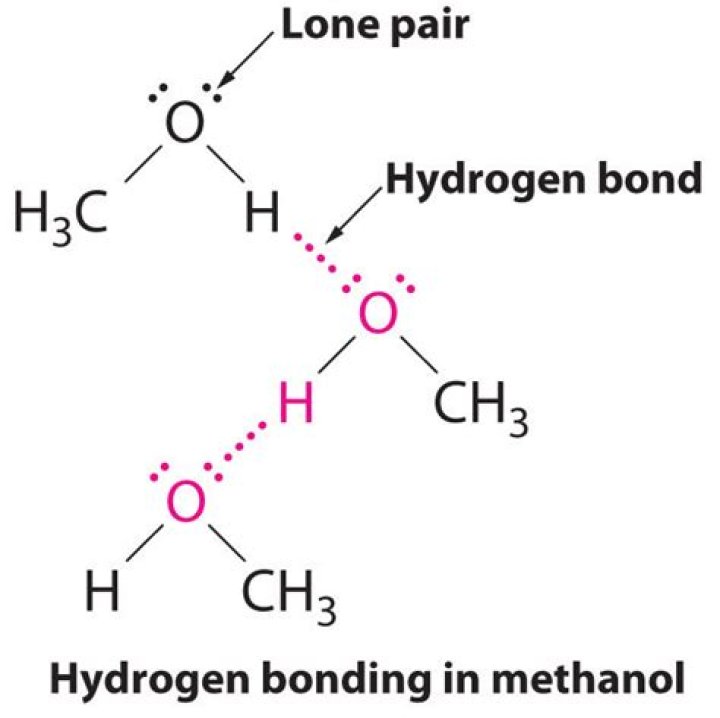
Only CH₃NH₂ and CH₃OH can have hydrogen bonds between other molecules of the same kind. To have hydrogen bonding, you need an N, O, or F atom in one molecule and an H attached to an N, O, or F atom in another molecule. See What is a Hydrogen bond?
Which type of bond is present in CH3OH?
Its a covalent bond. In CH3OH, the the OH group loses the H+ portion but leaves the electrons with Oxygen since oxygen is so electronegative.
How many hydrogen bonds does CH3OH have?
Methanol generally only forms three strong hydrogen bonds, two as proton acceptors (via the lone-pair electrons on oxygen) and one as a proton donor (Lee et al., 1988).
Ch3oh intermolecular forces has hydrogen bonding, dipole dipole attraction and London dispersion forces.
What type of bond does methanol have?
The molecule of methanol has covalent bonds within it. The hydroxyl group is connected with carbon with one of its four bonds. The shape of the molecule of methanol (CH3OH) is asymmetrical in shape.
N2: Nitrogen gas (N2) is diatomic and non-polar because both nitrogen atoms have the same degree of electro-negativity. London dispersion forces allows nitrogen atoms stick together to form a liquid. All molecules have dispersion forces.
What are the intermolecular forces found in CH3OH?
The common types of intermolecular forces of attraction that may exist for compounds such as methanol are hydrogen bonding, London Dispersion Force, or the dipole-dipole force of attraction.
What kind of intermolecular forces act between two methanol CH3OH molecules?
Methanol is polar, and will exhibit dipole interactions. It also contains the -OH alcohol group which will allow for hydrogen bonding.
There are 6 sigma bonds and 0 pi bonds in this molecule.
Does 1 propanol have hydrogen bonding?
The small alcohols ethanol, 1-propanol, and 2-propanol are miscible in water, form strong hydrogen bonds with water molecules, and are usually known as inhibitors for clathrate hydrate formation.
Is CH3OH a hydrogen bond donor and acceptor?
Methanol (CH3OH) is protic because it can donate a hydrogen for hydrogen bonding. Its oxygen atom is a hydrogen bond acceptor, the molecule has no hydrogen with enough δ+ charge to be a hydrogen bond donor.
Does CH3 2o have hydrogen bonding?
Both molecules possess dipole moments but CH3CH2OH contains hydrogen bonded to an electronegative element so H-bonding is possible. Both molecules possess dipole moments but CH3COOH contains hydrogen bonded to an electronegative element so H-bonding is possible.
In (CH3)3N ( C H 3 ) 3 N , the hydrogen atoms are bonded to carbon atoms. Carbon is not a very electronegative atom so it cannot act as a hydrogen donor. Although nitrogen is very electronegative and can act as a hydrogen acceptor, there are no hydrogens to accept.



