Is CH3F polar?
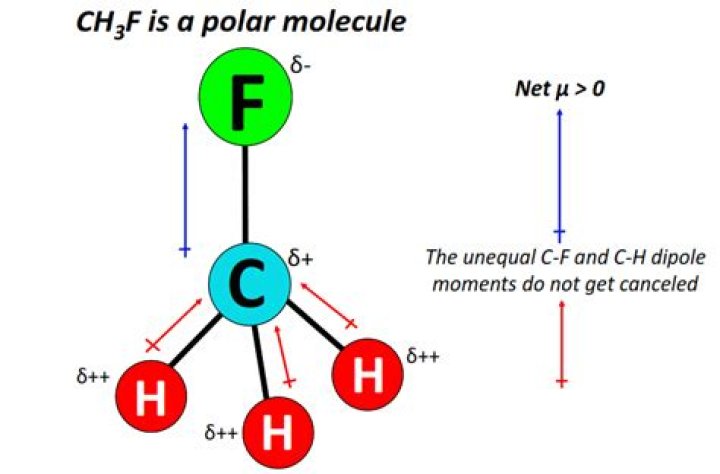
Is CH3F polar or nonpolar, then? CH3F (Fluoromethane) is polar due to its tetrahedral geometrical shape caused by the presence of a high electronegativity value fluorine atom.
Is CH3I polar?
CH3I is polar, because of the single iodine. Iodine is a somewhat electronegative atom, and it will pull some of the electrons of the slightly less electronegative carbon towards it.
Is CH3Cl polar?
Because the C-Cl bond is polar, the CH3Cl has a net dipole, so CH3Cl is polar.
Which molecule is non-polar?
Nonpolar Molecule Examples
Examples of homonuclear nonpolar molecules are oxygen (O2), nitrogen (N2), and ozone (O3). Other nonpolar molecules include carbon dioxide (CO2) and the organic molecules methane (CH4), toluene, and gasoline. Most carbon compounds are nonpolar.
What electronegativity is nonpolar?
Although there are no hard and fast rules, the general rule is if the difference in electronegativities is less than about 0.4, the bond is considered nonpolar; if the difference is greater than 0.4, the bond is considered polar.
Why is methyl fluoride polar?
The methyl fluoride CH3F is a polar molecule. The polarity is due to the difference in the electronegativity between fluorine and carbon. For such a molecule, the possible intermolecular interactions are: London Dispersion Force (LDF).
Which is more polar CHF3 or CH3F?
The dipole moment of CH3F is 1.81D whereas the dipole moment of CHF3 is 1.61D. If the hybridization and the bond lengths in both CH3F and CHF3 were the same, these two molecules would have the same dipole moment. This is NOT a nonpolar molecule!
What is the electronegativity of CH3I?
Calculating the electronegative difference in carbon and iodine in CH3I. The carbon atom has an electronegativity of 2.55, while iodine has an electronegativity of 2.96 in the CH3I molecule.
What shape is CH3I?
The CH3I molecule has a tetrahedral geometry shape because it contains one iodine and three hydrogen atoms. There are three C-H and one C-I bond at the CH3I molecular geometry. After linking the three hydrogens and one iodine atom in the tetrahedral form, it maintains the tetrahedral-like structure.
Is NCS polar or nonpolar?
Interestingly the Li and Na phases are noncentrosymmetric (NCS) and polar, whereas the K, Rb, Cs, and Tl analogues are centrosymmetric (CS) and nonpolar.
Why is CH3Cl more polar than CHCl3?
Dipole moment of CH3Cl is GREATER than CHCl3. Now, why? Because in case of CHCl3 the bond angle Cl-C-Cl is larger due to larger atoms(Cl) hence net dipole moment decreases.
Is Hcooh polar or nonpolar?
The molecule is polar.
Is non polar hydrophobic?
Nonpolar molecules are hydrophobic; “hydro-” means water and “-phobic” means fear. Nonpolar molecule are water fearing and do not easily dissolve in water.
What is meant by non polar?
Definition of nonpolar
: not polar especially : consisting of molecules not having a dipole a nonpolar solvent.



