Is CH2Cl2 polar or nonpolar?
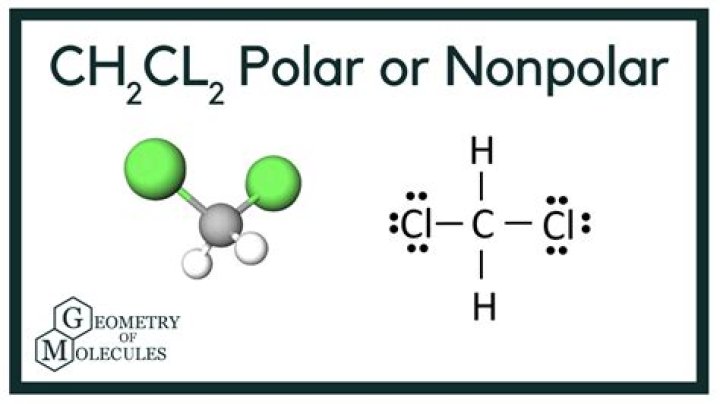
Though a Chlorine atom is nonpolar, the polar molecule emerges after valence electrons of nonpolar molecules bond its properties. So, it is indeed a fact that even if there are nonpolar molecules, but the bonds do not cancel, and the geometry is showing polarity, then CH2CL2 is polar.
Why CH2Cl2 is a polar molecule?
CH2Cl2 polar molecule The molecule is tetrahedral due to 4 electron repulsions around the central C atom. Although the bond arrangement around the C atom in CH2Cl2 is symmetrical, the differing polarities of the C–H and C–Cl bonds means the effect of the polar bonds is not cancelled, so the molecule is polar.
Is dichloromethane a polar solvent?
Dichloromethane (DCM or methylene chloride) is an organochloride compound with the formula CH2Cl2. This colorless, volatile liquid with a chloroform-like, sweet odour is widely used as a solvent. Although it is not miscible with water, it is polar, and miscible with many organic solvents.
Hydrogen=2.2, carbon=2.5 and chlorine=3.1. So, electronegativity difference between C-H=0.3 and C-Cl=0.6. It proves that CH2Cl2 is polar but a moderate polar as the difference between their electronegativity is quite small. The total number of valence electrons in the CH2Cl2 molecule is 20.
What shape is CH2Cl2?
The CH2Cl2 molecule is polar in nature. As the shape of the molecule is tetrahedral and Carbon and Chlorine have a difference in their electronegativity.
CH4 is a C surrounded by all H’s, so all of the H’s pull the electrons from the carbon the same amount (i.e. nonpolar). In CH2Cl2, the Cl pulls much more on the Carbon’s electrons (since it has higher electronegativity) than the Hydrogens, which sets up a dipole moment (i.e. polar).
What is the bond angle of CH2Cl2?
So for example methane, CH4 and dichloromethane, CH2Cl2, will have the same tetrahedral geometry and 109.5º bond angles since they both have four bonding pairs and no non-bonding pairs of electrons around the central carbon.
Does CCl2H2 have polar bonds?
Re: Polarity of CCl2H2
Since H and Cl have different electronegativities, the C-H and C-Cl dipole moments are also different. So although the 4 dipole moments do point in the same direction, they are not equivalent and thus do not cancel, making the overall molecule polar.
There are four electron groups around the central atom. Electron groups include lone pairs and atoms around the central atom: for CH2Cl2, there are 4 atoms, or a total of 4 electron groups around C. This corresponds to AX4 or tetrahedral. For CH2Cl2, its electron pair geometry is tetrahedral (AX4).
What is the dipole moment of CH2Cl2?
In CH2Cl2, the moment of two C-Cl bond is added at 109∘(approx.) so the resultant is greater than on C-Cl bond. In case of CH3Cl, the dipole moment of one C-Cl bond is added with moment of one C-H bond (resultant of three C-H bonds). But this should mean moment of CHCl3≈ moment of CH3Cl, but clearly this is not so.
Is dichloromethane more polar than water?
Yeh. CH2Cl2 is barely polar actually since C-Cl has deltaEN=0.5 where C-H gives a 0.4. Much less polar than water for sure..
Is CH2Cl2 planar?
Re: Atom arrangement of CH2Cl2
You would be right if CH2Cl2 had a square planar geometry, but its steric number is 4, not 6. Therefore you know it has a tetrahedral geometry, and if you pick any two places for Cl and H to be randomly, you would see it has a net dipole moment towards the side with the two H.
Organic solvents like dichloromethane are immiscible in water because water is, by contrast, a very polar solvent. When non-polar substances enter an aqueous solution, they exhibit hydrophobic behaviour by aggregating together and excluding the water molecules.
Which is the best description of the molecule dichloromethane CH2CL2?
Dichloromethane appears as a colorless liquid with a sweet, penetrating, ether-like odor. Noncombustible by if exposed to high temperatures may emit toxic chloride fumes.
Is dichloromethane soluble in water?
It is moderately soluble in water (2 g/100 ml at 20 °C) and soluble in most organic solvents such as ethanol, ether, phenols, aldehydes and ketones. Its evaporation rate is 27.5 (reference liquid is butyl acetate = 1). DCM vapours are heavier than air.



