Is CH2Cl2 more polar than water?
CH2Cl2 is barely polar actually since C-Cl has deltaEN=0.5 where C-H gives a 0.4. Much less polar than water for sure.. Well that explains it. Thanks.
How polar is CH2Cl2?
So, Is CH2Cl2 polar or nonpolar? CH2Cl2 is a polar molecule due to its tetrahedral geometrical shape and difference between the electronegativity of Carbon, Hydrogen and Chlorine atoms. This develops a dipole moment across C-Cl and C-H bonds and the entire molecule results in a net 1.67 D dipole moment.
Why is CH2Cl2 not soluble in water?
Organic solvents like dichloromethane are immiscible in water because water is, by contrast, a very polar solvent. Organic solvents, like ethyl acetate, are mostly less dense than water.
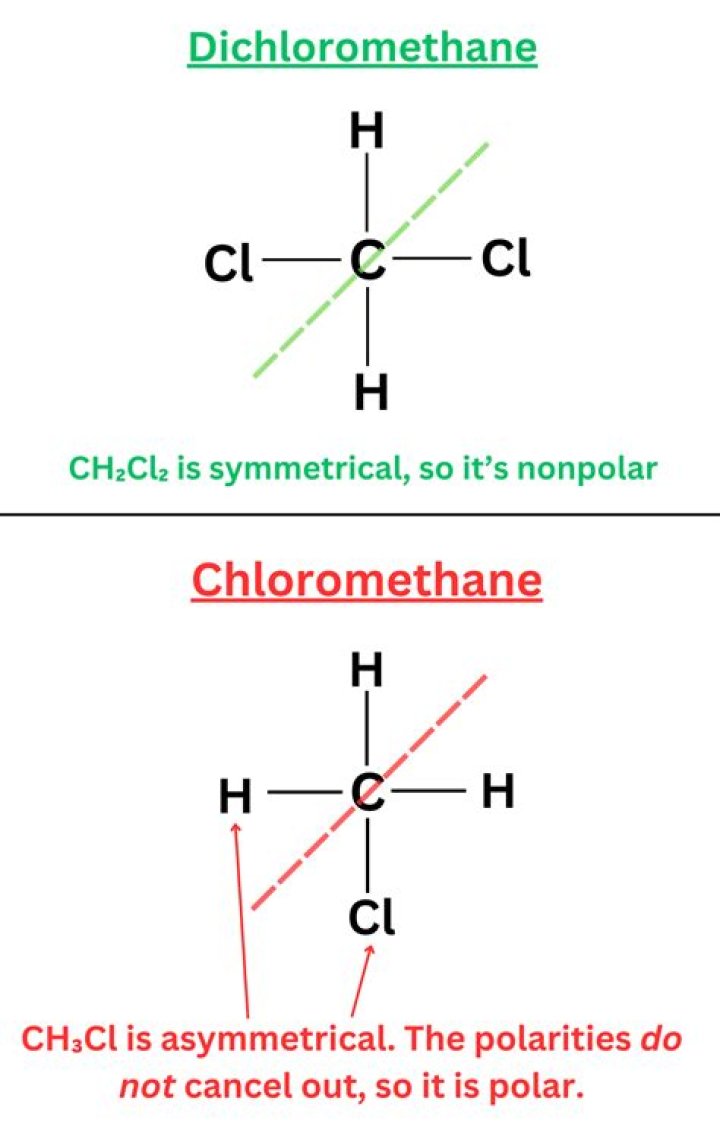
CH4 is a C surrounded by all H’s, so all of the H’s pull the electrons from the carbon the same amount (i.e. nonpolar). In CH2Cl2, the Cl pulls much more on the Carbon’s electrons (since it has higher electronegativity) than the Hydrogens, which sets up a dipole moment (i.e. polar).
Is dichloromethane very polar?
Dichloromethane is polar because it has different polarity bonds and its shape cannot arrange those bond dipoles to cancel out. Note that polarity can be considered for the whole molecule, functional group or particular bonds.
Water is a polar molecule and also acts as a polar solvent. When a chemical species is said to be “polar,” this means that the positive and negative electrical charges are unevenly distributed. The positive charge comes from the atomic nucleus, while the electrons supply the negative charge.
Why is water considered polar?
The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms. When a neutral molecule has a positive area at one end and a negative area at the other, it is a polar molecule.
Is PH3 polar or nonpolar?
PH3 is a polar molecule because it has a bent structure due to lone pairs of electrons and electron-electron repulsion. Phosphorus’s electronegativity is a nonpolar molecule because it is the same, but since Phosphorus has a lone pair, PH3 is a polar molecule.
Water is a Polar Covalent Molecule
The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).
Is h2o polar?
Water ( H2O ) is polar because of the bent shape of the molecule. It also acts as a polar solvent. The molecule is made up of two hydrogen atoms and one oxygen atom. When two water molecules get close together, the polar forces work to draw the molecules together.
Is dichloromethane more dense than water?
The density of the dichloromethane is greater than that of the water, therefore it sinks to the bottom.
Are hexane and dichloromethane miscible?
For example, dichloromethane and hexane mix together pretty well. In this case, the dipoles between dichloromethane are much smaller; they aren’t held back from the hexane molecules as strongly. On the other hand, the interaction between the the hexane and dichloromethane is actually amplified a little bit.
The C-C and C-H bonds in hydrocarbon molecules, such as ethane, C2H6, are not significantly polar, so hydrocarbons are non-polar molecular substances and hydrocarbon polymers like polyethylene or polypropylene are non-polar also. Generally polar polymers are more permeable to water than non-polar polymers.
Is CH2CL2 soluble?
It is moderately soluble in water (2 g/100 ml at 20 °C) and soluble in most organic solvents such as ethanol, ether, phenols, aldehydes and ketones.
How is dichloromethane removed from water?
Air stripping, adsorption and pervaporation were found to be effective in separating DCM from water with a process efficiency of about 99%, 90% and 80% respectively.
Is c6h12 soluble in water?
Cyclohexane appears as a clear colorless liquid with a petroleum-like odor. Used to make nylon, as a solvent, paint remover, and to make other chemicals. Flash point -4°F. Density 6.5 lb / gal (less than water) and insoluble in water.



