Is CBr4 polar or nonpolar?
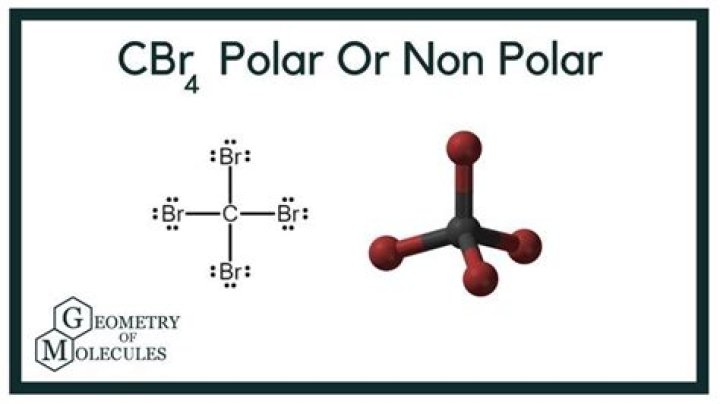
The CBr4 molecule is non-polar. Both CBr4 and CH3Br have four regions of electrons around the central carbon atom. These are all bonding electron regions (clouds) so the shape of both molecules is tetrahedral. The C-Br bond is polar due to the difference in electronegativity between C and Br.
What is CBr4 bond angle?
The Br-C-Br bond generates a 109-degree angle in a tetrahedral geometry. The CBr4 molecule has a tetrahedral shape because it contains four bromine atoms. The centre carbon atom of CBr4 has no lone pairs of electrons, resulting in tetrahedral electron geometry.
What kind of bond is CBr4?
The given compound carbon tetrabromide is a covalent compound.
Is CBr4 linear or bent?
CBr4 is in a tetrahedral molecular shape, so all the dipoles of the 4 polar C-Br bonds cancel each other out, resulting in an overall nonpolar molecule.
What is the correct Iupac name for CBr4?
Tetrabromomethane, CBr4, also known as carbon tetrabromide, is a carbon bromide. Both names are acceptable under IUPAC nomenclature.
How many lone pairs are in CBr4?
Carbon atoms have 4 valence electrons. With 4 Br, CBr4 will have 4 bonded pairs, no lone pairs.
How many electron groups does CBr4?
There are 12 electron groups (3 electron groups on each Br atom).



