Is calcium chloride covalent or ionic?
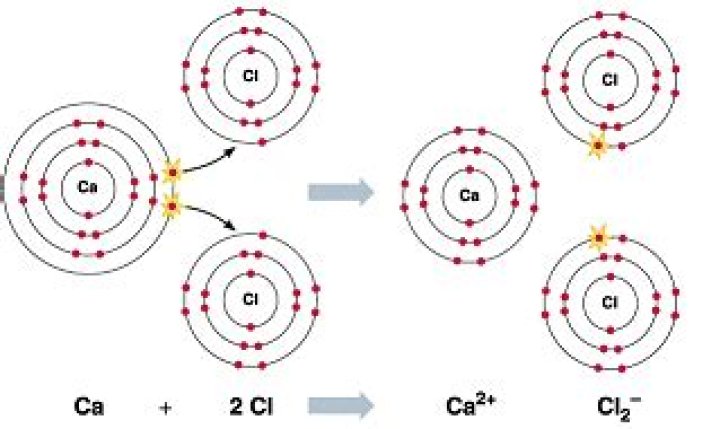
CaCl2 is an ionic bond. This is because the calcium gives up an electron to each of the chlorine atoms resulting in the calcium becoming Ca2+ ions…
Why calcium chloride is ionic?
Ionic compounds generally form between elements that are metals and elements that are nonmetals. For example, the metal calcium (Ca) and the nonmetal chlorine (Cl) form the ionic compound calcium chloride (CaCl2). Because the positive and negative charges cancel out, an ionic compound is neutral in charge.
How is calcium chloride ionic?
Calcium chloride is created from the ionic bonds that form between calcium cations and chloride anions. Calcium ions have a charge of +2, while chloride ions have a charge of -1. On screen is the chemical reaction for the formation of calcium chloride.
CaCl2 or Calcium Chloride is an ionic bond and not a covalent bond. Since there should be sharing of electrons between two atoms to be a covalent bonding.
Is calcium chloride an anion or cation?
Formula and structure: The chemical formula of calcium chloride is CaCl2, and its molar mass is 110.983 g/mol. It is an ionic compound consisting of the calcium cation (Ca2+) and two chlorine anions (Cl-). The bivalent calcium metal forms an ionic bond with two chlorine atoms, as shown below.
Therefore, each copper atom loses two electrons for two chlorine atoms. The result is a copper ion with a charge of positive two, and two chloride ions with a charge of negative one each. Hence, copper(II) chloride is an ionic compound.
Is N2O5 ionic or covalent?
N2O5 contains nitrogen and oxygen atoms, which, if you look at the periodic table, are both non-metals. When two non-metallic bonds form the covalent bonds. Regardless of whether NN, NO or O = O are formed, the bond is covalent.
What type of compound is calcium chloride?
Calcium chloride is an inorganic compound, a salt with the chemical formula CaCl2.
Increasing order of covalent character among given compounds is HgCl2
Is HgCl2 covalent?
HgCl2 is a triatomic molecule where the Hg atom is bonded with two Cl atoms. Here we see that formation of an ionic complex is taking place. And also, for the common ion effect to occur the compound must be an ionic compound to form ion after dissociation and here HgCl2 is a covalent compound.
What type of bond is CaCl2?
CaCl2 is an ionic bond. This is because the calcium gives up an electron to each of the chlorine atoms resulting in the calcium becoming Ca2+ ions



