Is CaCl2 Electrovalent compound?
In case of calcium chloride, the calcium gives up an electron to each chlorine atom, becoming Ca2+ ions and chlorine become Cl– ions. Step-by-step explanation: An ionic bond is formed between a metal and a nonmetal. … So in that regard, CaCl2 is ionic.
Which compound is Electrovalent?
The compounds which contain ionic or electrovalent bonds are Electrovalent or Ionic Compounds. Mainly electrovalent compounds are formed due to the reaction between highly electropositive and highly electronegative atoms.
Is CaCl is a Electrovalent bond?
CaCl2 will have electrovalent bonding because calcium is electropositive metal while chlorine is electronegative so they will combined with electrovalent bond.
Thus, we can see that calcium can lose two electrons and oxygen can accept two electrons. Thus, calcium gives its two valence electrons to oxygen and forms an electrovalent bond with oxygen, thus completing each other’s octet configuration as well. Thus, the correct option is B. Electrovalent compound .
Is CaCl2 a binary compound?
The name for CaCl2 is calcium chloride. This is a binary ionic compound, a metal and a non metal.
Calcium chloride is an ionic compound.
Is CaCl2 a covalent bond?
CaCl2 or Calcium Chloride is an ionic bond and not a covalent bond. Since there should be sharing of electrons between two atoms to be a covalent bonding. In case of calcium chloride, the calcium gives up an electron to each chlorine atom, becoming Ca2+ ions and chlorine become Cl– ions.
Is CaCl2 a metal or non metal?
The ions are held together by ionic bonds, which form an ionic compound. Ionic compounds generally form between elements that are metals and elements that are nonmetals. For example, the metal calcium (Ca) and the nonmetal chlorine (Cl) form the ionic compound calcium chloride (CaCl2).
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
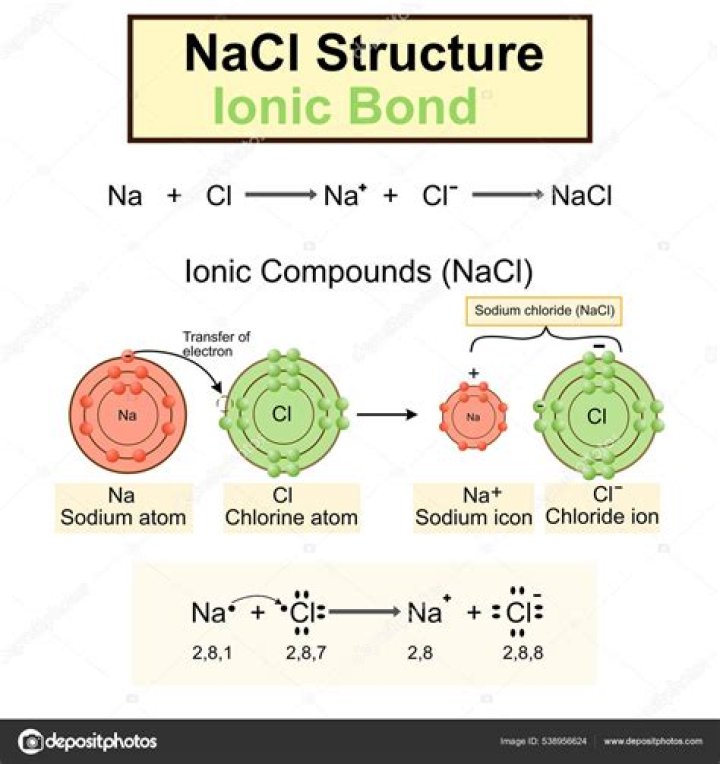
Is NaCl an Electrovalent compound?
It has seven electrons in its outermost shell or chlorine has 7 valence electrons. Since NaCl compounds are also formed by the transfer of one electron thus, NaCl is an electrovalent compound. Hence, NaCl is an electrovalent compound.
How is CaCl2 formed?
The formation of cacl2 happens mainly by reacting limestone (CaCo3) with hydrochloric acid (HCl). The chemical equation can be given as follows. It is also produced as a primary by-product while manufacturing the soda ash Na2CO3 using the Solvay process, where the limestone is reacted with NaCl solution.
What forces are holding the particles in CaCl2 together?
CaCl2 is held together by strong ionic (electrostatic) attractions between ions which require a much greater input of energy to overcome.
Because the bond forms between a metal (Ca) and a non-metal (O), the bond will most likely be ionic. If a bond forms between two non-metals, then the bond is most likely covalent.
Is calcium an oxide?
Calcium oxide, CaO, also known as lime or more specifically quicklime, is a white or grayish white solid produced in large quantities by roasting calcium carbonate so as to drive off carbon dioxide.
What type of bond is calcium oxide?
Calcium oxide is ionic because it is formed between a metal and non-metal and bonds formed between a metal and non-metal atoms are ionic. Determination of bonding in compounds: A bond formed between a metal and nonmetal atom is ionic.



