Is c2h5co2h polar or nonpolar?
C2H5COOH is polar, but C3H7OH is not polar.
Is it polar or non polar?
When things are different at each end, we call them polar. Some molecules have positive and negative ends too, and when they do, we call them polar. If they don’t, we call them non-polar. Things that are polar can attract and repel each other (opposite charges attract, alike charges repel).
Is propionic acid polar?
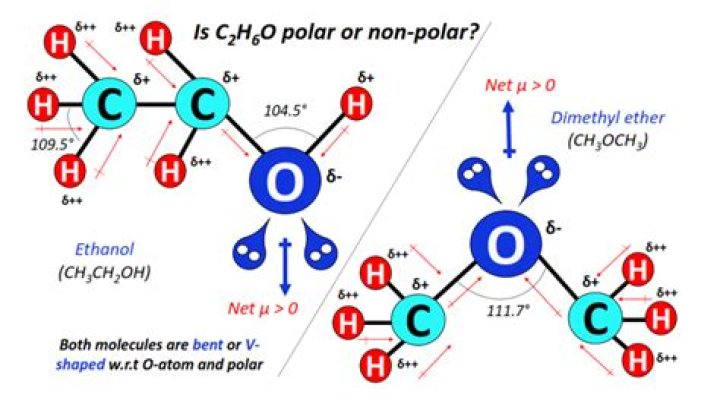
However, the polar tail wins in this round hence propanoic acid is polar in nature. Question: Is The Molecule CH3CH2OCH3 A Polar Or Nonpolar Molecule? 1 Answer.
C3H7OH is not polar, but C3H7OH is polar. Amino acids are acids because they always possess which functional group? A compound contains hydroxyl groups as its predominant functional group.
Which molecule is nonpolar?
Nonpolar Molecule Examples
Examples of homonuclear nonpolar molecules are oxygen (O2), nitrogen (N2), and ozone (O3). Other nonpolar molecules include carbon dioxide (CO2) and the organic molecules methane (CH4), toluene, and gasoline. Most carbon compounds are nonpolar.
Although there are no hard and fast rules, the general rule is if the difference in electronegativities is less than about 0.4, the bond is considered nonpolar; if the difference is greater than 0.4, the bond is considered polar.
Is propionic acid volatile?
Volatile fatty acids (VFAs)—mainly acetic acid, propionic acid, and butyric acid—have great importance as commodity chemicals, and biological production of these acids from various waste streams is gaining a huge amount of attention.
What is c2h5cooh?
The IUPAC name is propanoic acid.
Acetic acid(CH3COOH) is a polar molecule because it contains double-bonded oxygen which is more electronegative than a carbon atom, so, the difference of electronegativity in carbon and oxygen atom, generates a dipole moment in the C-O bond because of inducing a positive and negative charge on them.
Which solvent is non-polar?
Nonpolar solvents include alkanes (pentane, hexane, and heptane) and aromatics (benzene, toluene, and xylene). Other common nonpolar solvents include acetic acid, chloroform, diethyl ether, ethyl acetate, methylene chloride, and pyridine.
Is diethyl ether polar or nonpolar?
Dear Peter, Diethyl ether is considered as a non-polar solvent due to its low dielectric constant.
What intermolecular forces does c3h6o have?
Answer and Explanation: 1) Acetone is a dipolar molecule. Therefore, the dominant intermolecular forces between the acetone molecules are dipole-dipole interactions.
Hexane is a non-polar solvent with a boiling point of 68°C, and is therefore the solvent of choice for oil extraction from rice bran to yield rice bran oil (RBO).
Is sibr4 polar or nonpolar?
The electronegativity of silicon is 1.8, that of bromine is 2.8. The silicon – bromine bond is polar. Showing these bonds as arrows in a tetrahedral structure clarifies that silicon tetrabromide is a nonpolar molecule.
Is CH4 polar covalent?
So, is CH4 polar or nonpolar? CH4 is a nonpolar molecule as it has a symmetric tetrahedral geometrical shape with four identical C-H bonds. The electronegativity of carbon and hydrogen is 2.55 and 2.2, respectively, which causes the partial charges to be almost zero.
Why is propanol polar?
1-Propanol is a polar molecule. This is because it contains a O-H bond which is a polar covalent bond.
Examples of hydrophobic molecules include the alkanes, oils, fats, and greasy substances in general. Hydrophobic materials are used for oil removal from water, the management of oil spills, and chemical separation processes to remove non-polar substances from polar compounds.



