Is C2H4 polar or nonpolar?
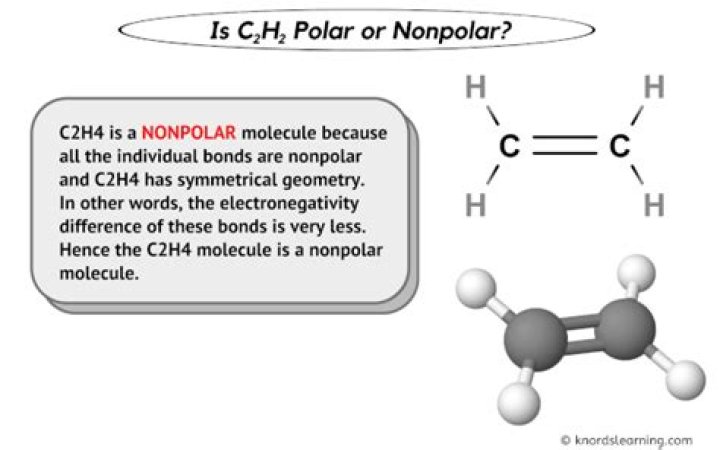
The hydrocarbons are mostly non-polar in nature because the electronegativity difference is not large between the carbon and hydrogen. Carbon and hydrogen atoms are bonded together by covalent bond. That’s why C2H4 C 2 H 4 is non-polar in nature. So, C2H4 C 2 H 4 is non-polar in nature.
Is C2H2 polar or nonpolar?
Answer: C2H2 ( acetylene) is nonpolar because of the nonpolar covalent bonds being arranged in a linear structure around the two central carbon atoms.
Is it polar or non-polar?
When things are different at each end, we call them polar. Some molecules have positive and negative ends too, and when they do, we call them polar. If they don’t, we call them non-polar. Things that are polar can attract and repel each other (opposite charges attract, alike charges repel).
By contrast, in ethene, C2H4, each H-C-H bond angle is 116.6° and each H-C-C bond angle is 121.7°. All six atoms of ethene lie in the same plane.
What is molecular geometry of C2H4?
The molecular geometry of C2H4 is trigonal planar and its electron geometry is also trigonal planar according to VSEPR (Valence shell electron pair repulsion theory).
A NON-polar, totally symmetrical molecule like C2Cl4, known as tetrachloroethene, is used for DRY CLEANING clothes because it attracts to the non-polar grease stains that are NOT effectively removed by polar water molecules. The dry cleaning process is NOT dry at all (C2Cl4 is a liquid).
Is C2H6O polar or nonpolar?
Ethanol, or C2H6O, has two different types of bonding between its constituent atoms. The bonds between the hydrogen and carbon atoms are nonpolar covalent bonds. The hydrogen-oxygen and carbon-oxygen bonds are polar covalent bonds.
Is C2H4 a linear molecule?
When we draw the Lewis Structure of C2H4, we find a linear 2-D representation. In reality, the molecular shape of ethene is not linear.
Yes ethene is a planar molecule because there is no free rotation around the carbon-carbon double bond. The molecule of ethene is a planar molecule.
What are the intermolecular forces of C2H4?
Intermolecular Forces : Example Question #1
Ethene is an organic molecule composed of two carbon atoms, joined by a double bond, and four hydrogen atoms. Ethene, like all molecules, exhibits London dispersion forces. This molecule, however, has no net dipole moment, so it will not exhibit dipole-dipole attraction.
Can C2H4 form hydrogen bonds?
For example, intramolecular hydrogen bonding occurs in ethylene glycol (C2H4(OH)2) between its two hydroxyl groups due to the molecular geometry. Intermolecular hydrogen bonds occur between separate molecules in a substance.
Which molecule is nonpolar?
Nonpolar Molecule Examples
Examples of homonuclear nonpolar molecules are oxygen (O2), nitrogen (N2), and ozone (O3). Other nonpolar molecules include carbon dioxide (CO2) and the organic molecules methane (CH4), toluene, and gasoline. Most carbon compounds are nonpolar.
Although there are no hard and fast rules, the general rule is if the difference in electronegativities is less than about 0.4, the bond is considered nonpolar; if the difference is greater than 0.4, the bond is considered polar.
Which chemical from the list are non polar?
Any of the homonuclear diatomic elements: H2, N2, O2, Cl2 (These are truly nonpolar molecules.) Carbon dioxide – CO. Boron trifluoride – BF. Benzene – C6H.



