Is C2H4 a shape?
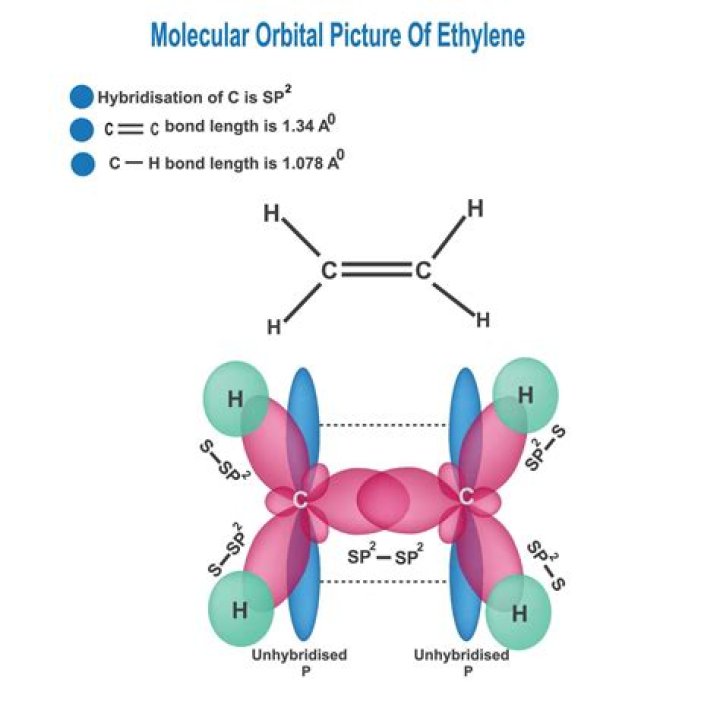
Ethylene, C2H4 has the Lewis Structure: The molecular shape is predicted to be trigonal planar around each carbon atom. This is composed of a σ framework and a π-bond.
What shape is C2H4?
Ethylene, C2H4 has the Lewis Structure: The molecular shape is predicted to be trigonal planar around each carbon atom. This is composed of a σ framework and a π-bond.
Is C2H4 trigonal?
The molecular geometry or shape of C2H4 is trigonal planar. Ethene lewis’s structure is made up of two carbon and four hydrogens, the carbon atoms at the middle position and the hydrogen atom at the surrounding position.
For C2H4. In C2H4, if we look into the lewis structure, we will see that there are three bonded pairs of electrons around each carbon and zero lone pair. According to the VSEPR chart, the shape of the ethene molecule is trigonal planar. This is due to the fact that each carbon surrounds a planar triangle.
What is the geometry of XeF2?
XeF2 molecular geometry is linear. It acquires such shape as the lone pairs present around the central atom tend to take up equatorial positions. The bond angle is said to be 180°.
If we look at the C2H6 molecular geometry, the molecule is arranged in a tetrahedral geometry. The central carbon atoms are surrounded by H-atoms with a bond angle of 109.5o.
Is C2H2 planar?
in C2H6 and CH4 C atoms are sp3 hybridised and thus a tetrahedral 3d shape forms or u can say that one bondind lies out of plane. C2H2 is linear in shape.
Is C2H2 nonpolar or polar?
Answer: C2H2 ( acetylene) is nonpolar because of the nonpolar covalent bonds being arranged in a linear structure around the two central carbon atoms.
C2H4 (Ethylene) consists of 2 carbon atom and 4 hydrogen atoms. The carbon-carbon bond is double covalent bond ie; sharing of two-electron pairs occurs.
Is C2H4 polar or nonpolar?
The hydrocarbons are mostly non-polar in nature because the electronegativity difference is not large between the carbon and hydrogen. Carbon and hydrogen atoms are bonded together by covalent bond. That’s why C2H4 C 2 H 4 is non-polar in nature. So, C2H4 C 2 H 4 is non-polar in nature.
What defines the shape of a molecule?
The shape of a molecule is determined by the location of the nuclei and its electrons. The electrons and the nuclei settle into positions that minimize repulsion and maximize attraction. Thus, the molecule’s shape reflects its equilibrium state in which it has the lowest possible energy in the system.



