Is BrCl3 a polar molecule?
BrCl3 is polar in nature and the bond angle is distorted from the ideal due to the presence of 2 lone pairs.
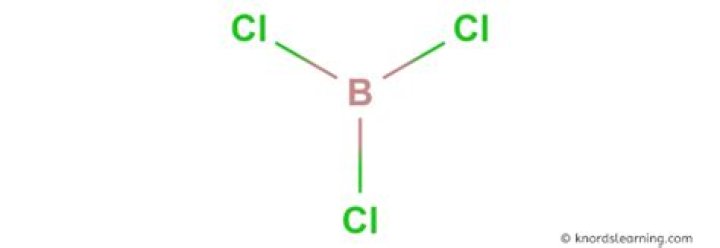
Is BCl3 polar or nonpolar molecule?
Boron Trichloride or BCl3 is a nonpolar compound because of its symmetrical structure ie; Trigonal Planar. The B-Cl bond itself is polar because of the difference in electronegativity of Boron(2.04) and Chlorine(3.16) atoms and all three B-Cl bonds lie at 120 degrees to each other.
What is BrCl3?
Bromine chloride (BrCl3)
If the arrangement is symmetrical and the arrows are of equal length, the molecule is nonpolar. If the arrows are of different lengths, and if they do not balance each other, the molecule is polar. If the arrangement is asymmetrical, the molecule is polar.
Is BCl3 polar covalent bond?
BCl3 B C l 3 has trigonal planar geometry. There are three polar B-Cl bonds in this compound but due to the symmetry in its structure, all the bond dipoles of polar bonds cancel each other resulting in a zero resultant dipole moment of the molecule. So, BCl3 B C l 3 is polar.
BCl3 is a trigonal planar molecule like the other boron trihalides, and has a bond length of 175pm.
Is BrCl3 a molecular or ionic compound?
BrCl3 is an interhalogen molecule with a molecular weight of 186.26g/mol. Interhalogen molecules are those which contain two or more halogen atoms and no atoms of any other groups. A number of the interhalogen compounds are unstable and are mostly covalent in nature.
Is clbr3 polar or nonpolar?
The molecular geometry of ClBr3 is j) T-shaped with a bond angle of c) 120°. This means ClBr3 is b) nonpolar.
Examples of polar molecules include:
Water – H2O.Ammonia – NH. Sulfur dioxide – SO. Hydrogen sulfide – H2S.Carbon monoxide – CO.Ozone – O. Hydrofluoric acid – HF (and other molecules with a single H)Ethanol – C2H6O (and other alcohols with an OH at one end)
What are examples of polar and nonpolar molecules?
Solution
Water is polar. Any molecule with lone pairs of electrons around the central atom is polar.Methanol is polar. Hydrogen cyanide is polar. Oxygen is nonpolar. Propane is nonpolar, because it is symmetric, with H atoms bonded to every side around the central atoms and no unshared pairs of electrons.
Which of the following is non-polar molecule?
Any of the homonuclear diatomic elements: H2, N2, O2, Cl2 (These are truly nonpolar molecules.) Carbon dioxide – CO. Benzene – C6H. Carbon tetrachloride – CCl.
What makes a molecule polar?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A dipole is any molecule with a positive end and a negative end, resulting from unequal distribution of electron density throughout the molecule.
Asymmetrical molecules are always polar. Symmetrical molecules can be polar and non-polar. Polarity is dependent on the difference in electronegativity of the molecule’s atoms and shape. The atom with the largest electronegativity will attract the electrons more frequently than the other atoms in the molecule.
Is IF6+ polar or nonpolar?
The polarity of IF6 is polar. Iodine heptafluoride, also known as iodine(VII) fluoride or iodine fluoride, is an interhalogen compound with the chemical formula I F 7.
Does BCl3 have a dipole?
BCl3, for example, has no dipole moment, while NH3 does. This suggests that in BCl3 the chlorines around boron are in a trigonal planar arrangement, while the hydrogens around nitrogen in NH3 would have a less symmetrical arrangement (e.g., trigonal pyramidal, T-shaped).
What is the polarity of AsH3?
The correct option of the following question is b. AsH3 is a polar compound. Arsenic trifluoride has a dipole moment throughout its structure, therefore, this compound is polar in nature.
Conclusion. Phosphorus trichloride is a chemical compound having 3 chlorine atoms and 1 Phosphorus atom with one lone pair on Phosphorus atom. Due to asymmetric shape and difference between the electronegativity, the PCl3 molecule is polar in nature.



