Is BH3 a dipole?
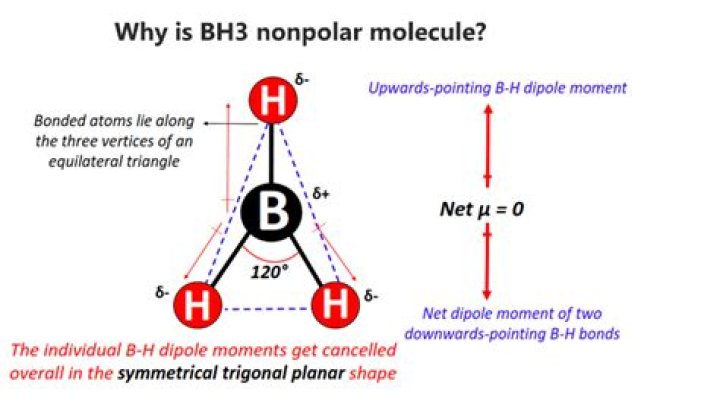
Borane gas molecule consists of a single boron atom surrounded by three hydrogen atoms. … The polarity of B-H bonds gets canceled out by each other due to the symmetric shape of the molecule. As a result, the overall dipole moment of the BH3 molecule turns out to be zero. Due to these reasons, BH3 is nonpolar.
What is the dipole moment of BH3?
In tetra-atomic boron trihydride (BH3), the dipole moment is zero, but that of ammonia (NH3) is 1.49D. This is because BH3 has a symmetrical structure and the 3 B-H bonds are placed at an angle of 120 degrees to each other.
Why does BF3 not have a dipole moment?
Boron trifluoride is has zero dipole moment although it has three polar B—F bonds. This is because BF3 has sp2 hybridisation and regular trigonal planal geometry. And hence the individual dipole moments of polar bonds get cancelled and overall dipole moment is zero.
Yes. Although NH3 shows dipole-dipole intermolecular forces, the highly electronegative atoms resulted in polarity.
Does CO2 have a dipole moment?
Polar molecules have a non-zero net dipole moment. Both CO2 and H2O have two polar bonds. However the dipoles in the linear CO2 molecule cancel each other out, meaning that the CO2 molecule is non-polar. The polar bonds in the bent H2O molecule result in a net dipole moment, so H2O is polar.
The observed structure of the borane molecule, BH3, suggests sp2 hybridization for boron in this compound. The molecule is trigonal planar, and the boron atom is involved in three bonds to hydrogen atoms (Figure 7).
What type of chemical bond is BH3?
The boron atom in BH3 has 6 valence electrons. Consequently it is a strong Lewis acid and reacts with any Lewis base, L to form an adduct. in which the base donates its lone pair, forming a dative covalent bond.
Is BF3 ionic or covalent?
BF3 is a molecule consisting of an sp2 hybrid of Boron covalently bonded with 3 atoms of fluorine. The covalent bond tells us that electrons are shared, rather than lost by boron and gained by fluorine.
Boron trifluoride (BF3) has no dipole moment (mu = 0D) .
What intermolecular forces are present in BF3?
As a result, the only type of intermolecular forces in BF3 would be the London dispersion forces.
Are BH3 bonds polar?
So, Is BH3 Polar or Nonpolar? BH3 (Borane) is a nonpolar substance because of the symmetrical trigonal planar geometry. Also due to symmetric shape, the possibility of the polarity of B-H bonds cancels out by each other resulting in a nonpolar molecule.
Is BF3 polar?
Boron trifluoride (BF3) is a nonpolar molecule, whereas ammonia (NH3) is a polar molecule.
Covalent molecules made of only one type of atom, like hydrogen gas (H2), are nonpolar because the hydrogen atoms share their electrons equally. 3-6 for examples of polar and nonpolar molecules.
What are molecular dipoles?
Molecular dipoles. These occur when two atoms in a molecule have substantially different electronegativity: One atom attracts electrons more than another, becoming more negative, while the other atom becomes more positive. A molecule with a permanent dipole moment is called a polar molecule.
How do you find dipoles?
To estimate the dipole moments for each bond, it is common to use electronegativity difference between the bonded atoms as a rough gauge of the dipole strength. Subtract the two values, and point the negative end of the bond dipole toward the atom of greater electronegativity.
Is CO2 dispersion only?
Humaam H. Carbon Dioxide ( CO2 ) has covalent bonds and dispersion forces. Thus, although CO₂ has polar bonds, it is a nonpolar molecule. Therefore, the only intermolecular forces are London dispersion forces.
BF3 is an sp2 hybridization. It is sp2 for this molecule because one π (pi) bond is needed for the double bond between the Boron, and just three σ bonds are produced per Boron atom. The atomic S and P – orbitals in Boron outer shell mix to form three equivalent hybrid orbitals of sp2.
What type of atom is BF3?
BF3 has a boron atom with three outer-shell electrons in its ground state and three fluorine atoms containing seven outer electrons.
What is the bond angle of bh3?
Shape of BH3 is trigonal planar, bond angle is 120 degree.



