Is BA OH 2 ionic or molecular?
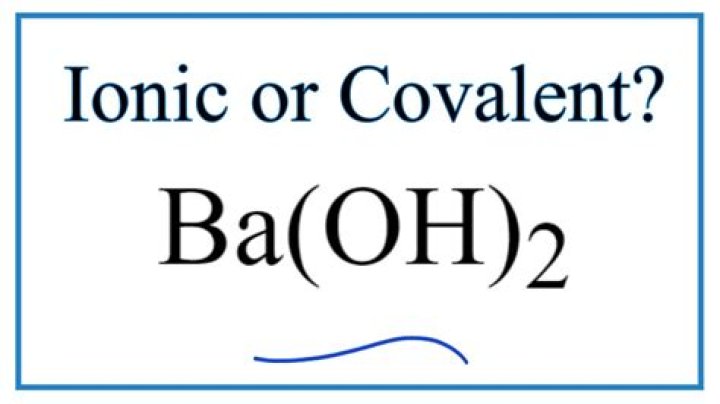
It is ionic in nature for example, Ba(OH)2 (barium hydroxide) in aqueous solution can provide two hydroxide ions per molecule.
Is hbro4 an ionic or molecular compound?
The compound perbromic acid is the inorganic compound with the formula HBrO4. It is an oxoacid of bromine. Perbromic acid is unstable and cannot be formed by displacement of chlorine from perchloric acid, as periodic acid is prepared; it can only be made by protonation of the perbromate ion.
What is NaBrO2?
Sodium bromite | NaBrO2 – PubChem.
Is NaBrO3 acid or base?
Sodium bromate, the inorganic compound with the chemical formula of NaBrO3, is the sodium salt of bromic acid. It is a strong oxidant.
What type of bond is CaO?
CaO is an ionic bond. Definition: Two-element compounds are usually ionic when one element is a metal and the other is a non-metal.
What is Cu2CO3?
Copper(I) Carbonate. Alias: Cuprous Carbonate. Formula: Cu2CO3.
Is NaClO4 ionic or molecular?
Yes NaClO4 has both ionic and covalent bond in it. Na is an atom which loses one electron to become Na+ ion and ClO4- is a poly atomic ion in which four oxygen atoms are covalently bonded with a Chlorine atom having charge of -1 on it and further more they combined together to form sodium chlorate.
Is KClO4 an acid or base?
(b) Potassium perchlorate, KClO4, is a neutral salt. Neither K+ nor ClO4- has any tendency to donate or accept a proton in dilute aqueous solutions. The reaction between a strong base KOH and the strong acid HClO4 produces KClO4. (c) Potassium hydrogen sulfate (also called potassium bisulfate) is an acidic salt.
Is hbro3 ionic or molecular?
Bromic acid, also known as hydrogen bromate, is an oxoacid with the molecular formula HBrO3.
What is HBrO4 called?
Perbromic acid | HBrO4 – PubChem.
What ion is BrO2?
Bromite | BrO2- – PubChem.
What is NaIO2?
Sodium Iodite NaIO2 Molecular Weight — EndMemo.



