Is all of methanol polar?
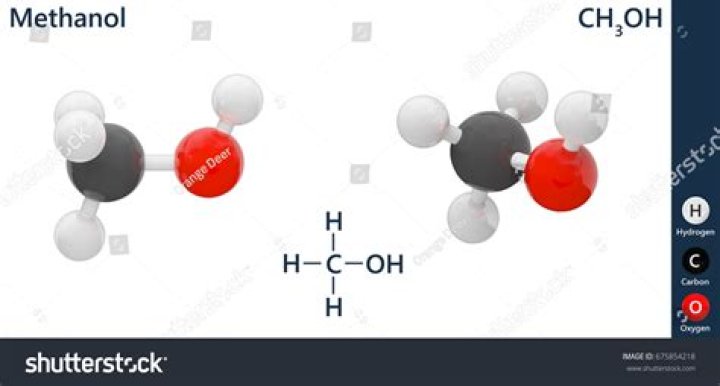
Yes. To begin with, methanol, H3C−OH , is asymmetrical, so it could not be nonpolar (being 100% symmetrical in all directions means all dipole moments would cancel out completely). … Therefore, there is a net dipole with the negative end pointing through oxygen, and methanol is polar.
Why is CH3OH polar?
CH3OH cannot be non-polar because there is a difference in electric charges among the atoms in the methanol molecule. Oxygen has more electron density due to its two lone pairs of electrons. This causes a net dipole pointing towards the Oxygen atom, making CH3OH polar. Thus CH3OH is a polar molecule.
Is methanol polar in water?
Methanol 100% is a good organic solvent to extract most of the compounds but it is not that polar compared to water. So combination of methanol-water will be good for most of the compounds from polar-non polar.
CH3F is a polar molecule, even though the tetrahedral geometry often leads to nonpolar molecules.
Is methanol or propanol more polar?
Propanol is a about equal polar and non-polar molecule:
When compared to the methanol, the total of three carbons and and multiple hydrogens it begins to show quite a bit more non-polar properties with larger areas of gray.
c. The oxygen in methanol is involved in two polar bonds, so we will focus on the geometry about this atom. The geometry about the oxygen (O) in methanol is tetrahedral (four groups of electrons).
Is O3 polar?
O3 is a polar molecule and it is due to its bent molecular geometry. In O3, the electric dipole moments of the bonds don’t counterbalance one another which results in a net dipole moment.
Is methanol polar covalent?
Methanol is polar in nature due to the unequal charge distribution of the atoms and its asymmetric molecular geometry. The oxygen is more electronegative than hydrogen and carbon gain partial negative charge and hydrogen and carbon gets a partial positive charge.
The hydrogen bonding between the water and methanol is as follows: Therefore, the methanol is soluble in water due to hydrogen bonding between methanol and water molecules.
How does methanol dissociate in water?
Methanol is soluble in water. Like water, methanol has relatively strong hydrogen bonding acting between its molecules. The forces of attraction between the ions and water molecules are strong enough to overcome the ion-ion and water-water force of attraction.
Why is methyl fluoride polar?
The methyl fluoride CH3F is a polar molecule. The polarity is due to the difference in the electronegativity between fluorine and carbon. For such a molecule, the possible intermolecular interactions are: London Dispersion Force (LDF).
Is H2 polar?
Covalent molecules made of only one type of atom, like hydrogen gas (H2), are nonpolar because the hydrogen atoms share their electrons equally. 3-6 for examples of polar and nonpolar molecules.
CH3F is a polar molecule due to the presence of higher electronegative Fluorine atom and gains a partial negative charge and other atoms gain partial positive charge and make the molecule polar. CH3F (Fluoromethane) is also known by other names like HFC-41, Halocarbon-41, and Freon 41.



