Is AlBr3 an ionic compound?
Aluminum bromide is an ionic compound, one where charged ions stick together due to electrostatic attraction that is formed from the reaction of aluminum with liquid bromine. Aluminum atoms give up three electrons resulting in Al+3, and bromine atoms gain one electron each resulting in Br-1.
What is AlBr3 used for?
Aluminum bromide (AlBr3) represents an important compound used in manufacturing and in the sciences. In both the standard chemistry classroom and in industries that rely upon the synthesis of other chemical compounds, aluminum bromide is often used as an acid catalyst in the synthesis of organic compounds.
Why is AlBr3 not an ionic compound?
CBr4 and AlBr3 are both not ionic compounds. AlBr3 might seem as though it should be one, but it has a low melting and boiling point and consists of Al2Br6 molecules, not ions.
Is AlBr3 polar covalent?
The dipole moments of each of the Al-Cl bond is directed at 120 degree angles to each other in a plane, and is therefore cancelled out. Therefore it is a non-polar molecule.
What type of solution is AlBr3?
Aluminum bromide will form an acidic solution.
The salt is formed from the reaction of a weak base and a strong acid. Salts formed from this combination of reactants result in an acidic salt. The chemical equation below shows the formation of the acidic salt.
What is the binary ionic compound of AlBr3?
The name of the ionic compound AlBr3 is aluminum bromide.
Is AlBr3 aqueous?
Aluminum Bromide is a highly water soluble crystalline Aluminum source for uses compatible with Bromides and lower (acidic) pH. Most metal bromide compounds are water soluble for uses in water treatment, chemical analysis and in ultra high purity for certain crystal growth applications.
Is AlBr3 is Lewis acid or Lewis base?
Here, AlBr3 A l B r 3 is a Lewis acid as it has an incomplete octet and
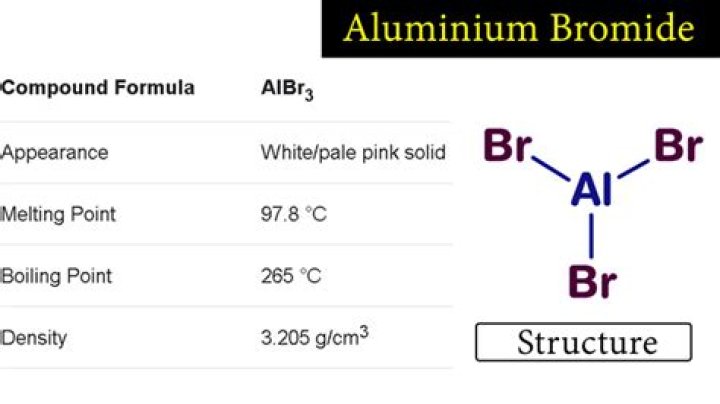
What is the the shape molecular geometry of AlBr3?
Hence geometry ( i.e. Electronic as well as molecular geometry ) of AlBr3 is Trigonal Planar.
What is al2s3 name?
CAS Number: 1302-81-4. Chemical Name: Aluminum sulfide.
Is AlBr3 a good conductor?
(a) Aluminium bromide exists as a dimer, Al2Br6. Due to the lack of free electrons, molten aluminum bromide is a poor conductor of electricity.
Is HCl ionic or covalent?
In its gaseous state, HCl is a covalently bonded molecule but exhibits considerable polarity. In aqueous solution, the molecules ionizes to produce H+ and Cl- in The presence of water acting as a Base. The ability to ionize also makes it useful as an electrolyte.
Is CaS ionic or covalent?
In terms of its atomic structure, CaS crystallizes in the same motif as sodium chloride indicating that the bonding in this material is highly ionic. The high melting point is also consistent with its description as an ionic solid.



