How many unpaired electrons V has?
So, there are 4 unpaired electrons. So, there are 3 unpaired electrons.
How do you find the number of unpaired electrons?
For finding the number of unpaired electrons, then first we have to find the atomic number of the element then write the configuration in the ground state, then according to the oxidation state subtract the number of electrons from the outer shell. So, there are 4 unpaired electrons.
How many unpaired electrons does V 2 have?
V(2+) will be (1s)2(2s)2(2p)6(3s)2(3p)6(4s)0(3d)3, and it will still have three unpaired electrons.
How many electrons are present in a V+ ion?
What is the electronic configuration of V+ (Vanadium with 22 electrons)? – Quora. Electrons enter the 3d subshell for the first time in Sc (Z= 21) and thereafter as at. no. increases, number of electrons in the 3d subshell also increases.
Does Al have 3 unpaired electrons?
As each orbital can hold a maximum of two electrons, the p orbital of the aluminum contains one unpaired electron. Therefore, the total number of unpaired electrons present in aluminum is 1.
What are the valence electrons of V?
Vanadium has five valence electrons.
How many valence electrons does v+?
This electron configuration of vanadium ion(V2+) shows that vanadium ion has three shells and the last shell has eleven electrons. For this, the valence electrons of the vanadium ion(V2+) are eleven.
How do you find unpaired electrons in O2?
O2Has two bonds, one is sigma bond and other is pi-bonded. The energy diagram of O2molecule is: The electrons in π∗2Px and π∗2Py remain unpaired. So, there are two unpaired electrons in O2.
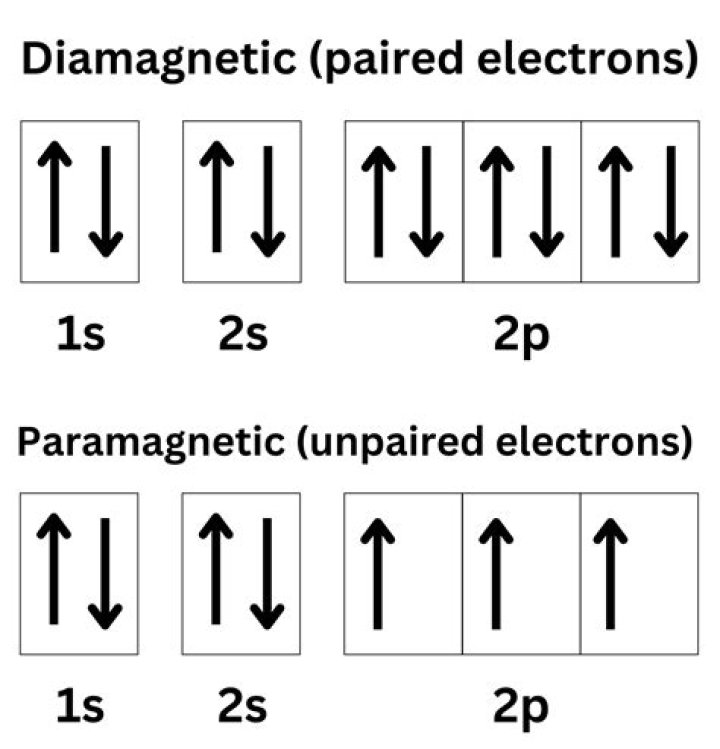
What are unpaired electrons called?
● Atoms with unpaired ↑ electrons are called paramagnetic.
Does Neon have unpaired electrons?
The number of unpaired electrons in the last orbit of an element is the valency of that element. The electron configuration of neon(Ne) shows that neon is an inert element. There are eight electrons in the last orbit of a neon atom. The neon atom has no unpaired electrons.



