How many unpaired electrons does cr2+ have?
Re: How does Cr2+ has 4 unpaired electrons? Answer: The electron configuration for Cr is [Ar]3d6 4s2 (note that 4s is higher in energy than 3d – that is important). Cr2+ means we remove 2 electrons, and we remove them from the highest energy level, which is 4s, so the electron configuration for Cr2+ is [Ar]3d6.
How many unpaired electrons does Cr2+ contain?
Re: How does Cr2+ has 4 unpaired electrons? Answer: The electron configuration for Cr is [Ar]3d6 4s2 (note that 4s is higher in energy than 3d – that is important). Cr2+ means we remove 2 electrons, and we remove them from the highest energy level, which is 4s, so the electron configuration for Cr2+ is [Ar]3d6.
How many unpaired electrons does chromium Cr have?
The free gaseous Cr atom has six unpaired electron.
∴ Number of unpaired electrons present is chromium is 6.
How do you find unpaired electrons?
For finding the number of unpaired electrons, then first we have to find the atomic number of the element then write the configuration in the ground state, then according to the oxidation state subtract the number of electrons from the outer shell. So, there are 4 unpaired electrons. So, there are 3 unpaired electrons.
Rubidium atoms have 37 electrons and the shell structure is 2.8. 18.8. 1.
Why is cr2+ reducing and mn3+ oxidizing?
Cr2+ is reducing agent as its configuration changes from d4 to d3, when it is oxidized to Cr3+ . On the other hand, the reduction of Mn3+ to Mn2+ results in the half-filled (d5) configuration which has extra stability hence Mn3+ acts as oxidizing agent.
Is cr2+ paramagnetic or diamagnetic?
Since there are 5 3d orbitals, they are not all at least singly filled yet, and thus, all three electrons in the lowest-energy configuration are unpaired. So, this is paramagnetic.
Therefore, both Si and S have 2 unpaired electrons.
How many electrons does P 3 have?
Phosphide is the anionic form of the element phosphorus. This element has an atomic number of 15. When neutral, it also contains 15 electrons.
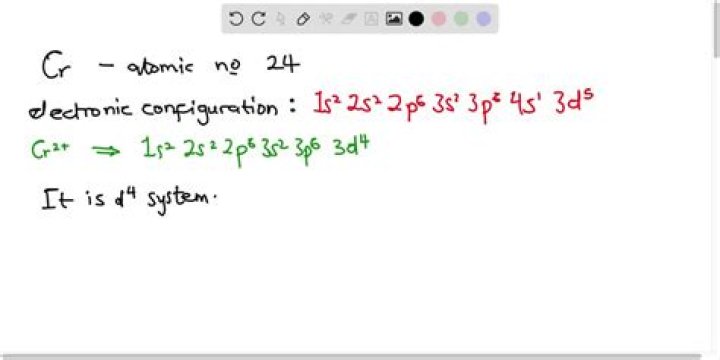
How do you find the unpaired electrons in chromium?
The ground-state electron configuration of chromium (Cr) atom is [Ar]4s13d5 [ A r ] 4 s 1 3 d 5 . There is 1 unpaired electron in the 4s subshell. Also, there are five electrons in the 3d subshell.
What is the meaning of unpaired electron?
An unpaired electron is an electron that occupies an orbital of an atom by itself, rather than as part of an electron pair.
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
How many protons neutrons and electrons are in CO?
Cobalt atoms have 27 electrons and 27 protons with 32 neutrons in the most abundant isotope.



