How many geometrical isomers are possible for Ma2b2c2?
Total number of stereoisomers possible for [Ma2b2c2] is Six (6) among which five are geometrical isomers. Among five geometrical isomers (diastereoisomers), one is optically active. Firstly, three pairs of ligands may be either trans to each other or cis to each other.
How many geometrical isomers are possible for Ma2b2c2?
There will be a total of 5 geometrical isomers formed.
Does Ma2b2c2 show optical isomerism?
The cis isomer shown in d is optically active so it exists as d and l isomer . This ,means that a complex of this type has 7 isomers out of which 5 are geometrical and 2 are optical isomers.
How many geometrical isomers can be formed from Ma2b2c2 where a B and C are monodentate ligands?
The octahedral complexes of [Ma2b2c2] type can exist in five geometrical isomers. The five geometrical isomers for the complex ion [PtCl2(NH3)2 (py)2]2+ are shown below.
How many geometrical isomers are formed by MA2BCDE?
8.12 Draw all possible isomers for [MA2BCDE]? Ignoring optical isomers, nine isomers are possible. Including optical isomers, 15 isomers are possible!
How many geometrical isomers are possible for ma3bcd?
Explanation: Octahedral MA2B2C2 type has total five geometrical isomers.
How many geometrical isomers are possible for MA3B3?
For an octahedral complex of the type, MA3B3, two isomers are possible (Figure 16). In one, the three same ligands (B), occupy opposite triangular faces of the octahedron; this is called the fac- isomer (for facial).
Is co Gly 3 shows optical isomerism?
Cr(gly)3 exhibit geometrical isomerism optical isomerism. It as cis and trans isomers.
How many geometrical isomers are possible for MA3B2C?
There are three possible cases in this combination MA3B2C. 3 A’s are in both cis and trans positions. Both A and B are in trans positions simultaneously.
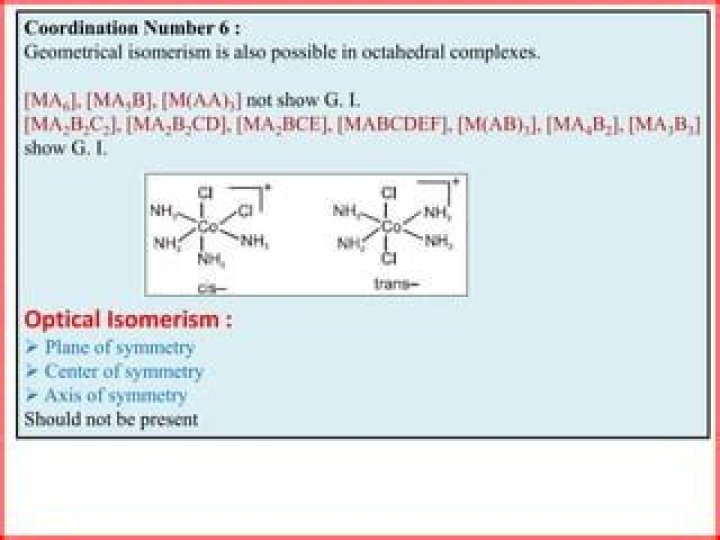
Which of the following do not show geometrical isomerism assume all ligands are Unidentate?
Complexes of MX6 and MX5L type (X and L are unidentate) do not show geometrical isomerism.
How many geometrical isomers are possible for the complex Mabed?
Thus, only three isomers are possible for the $text{ MABCD }$type of complex. The geometrical isomers are not possible in tetrahedral complexes as all the positions are adjacent to each other in these complexes.
How many geometrical isomers are possible for Mabcd?
A square-planar complex with formula [MABCD] can form three isomers. These isomers are called “A trans to B” and so on. Draw and label the three possible isomers.
How many geometrical isomers are possible for PT no2 )( nh3 )( nh2oh )( py?
4 isomers (Geometrical)
How many geometrical isomers are possible in a complex of type ma2 D 2 where A is monodentate and D is Bidentate?
For explanation I would say: In a complex of such type, the two A ligands can be arranged either adjacent to or opposite each other to form cis and trans isomers respectively, making it a total of two possible isomers.
Does ma4bc show geometrical isomerism?
Answer : This is a geometrical isomerism or cis-trans isomerism. Answer : This is a geometrical isomerism or cis-trans isomerism.
Does Ma3b3 show optical isomerism?
(ii) Optical isomerism: Optical isomers are those isomers which are non superimposable mirror images. Fac and Mer isomers : This type of geometrical isomerism occurs in octahedral coordination entities of the type [Ma3b3] like [Co(NH3)3(NO2)3].
What is facial and meridional isomers?
Facial and meridional isomers are geometrical isomers. These isomers are present in an octahedral ligand having two different types of monodentate ligands. Three ligands of one type and three ligands of other types are present. In the meridional isomer, the ammonia ligands are present on different faces of octahedrons.



