How many electrons dots does krypton have?
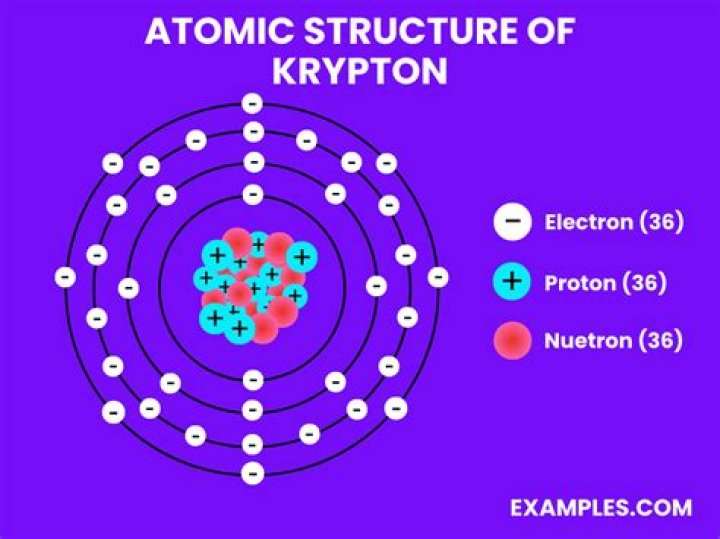
Since krypton is in the far right row of the periodic table, its outermost shell is full with eight electrons. This is one of the happy elements and has an electron configuration of 2-8-18-8. The other inert gases including argon and xenon also have full outer shells with eight electrons.
Why does krypton have 18 electrons?
A full M-shell (3rd shell) is 3s2 3p6 3d10. It holds 2 electrons in the s suborbital, 6 in a p suborbital, and 10 in a d suborbital. 2+6+10 = 18 electrons in a full shell.
How many bonds does krypton have?
Krypton Chemistry
Krypton can make 2 bonds to fluorine, as does argon. The molecule KrF2 has 2 covalent bonds between krypton and fluorine atoms. After 2 electrons in different 4p orbitals are excited to 4d orbitals, krypton can make 4 bonds to fluorine.
How many valence electron does krypton have?
Explanation: Since Kr or Krypton is a noble gas, it has a full valence shell or octet of 8 electrons.
What is the Valency of Zr?
During compound formation, the zirconium atom donates or share four electrons to another atom and the oxidation state of zirconium(Zr) is +4. Therefore, the valency of the zirconium element is 4.
How many electrons does Zr 4 have?
Zirconium has 40 electrons and 40 protons, so this picture has 40 electrons separated into the different levels.



