how does steepness of concentration gradient affect diffusion, check these out | Why does a higher concentration gradient affect diffusion?
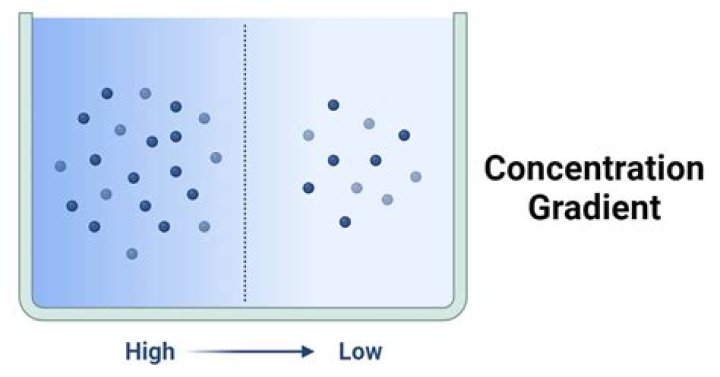
The bigger the difference, the steeper the concentration gradient and the faster the molecules of a substance will diffuse. The direction of diffusion is said to be ‘down’ or ‘with’ the concentration gradient. Diffusion stops when the concentration of the substance is equal in both areas.
Why does a higher concentration gradient affect diffusion?
The greater the difference in concentration, the quicker the rate of diffusion. The higher the temperature, the more kinetic energy the particles will have, so they will move and mix more quickly. The greater the surface area, the faster the rate of diffusion.
How is concentration gradient related to diffusion?
The concentration gradient therefore represents the concept that, just as a ball rolls down a slope, during diffusion molecules move down the concentration gradient. Higher concentration gradients will result in higher rates of diffusion. As the molecules move the gradient evens out until equilibrium is reached.
How does gradient size affect diffusion?
In passive transport, substances simply move from an area of higher concentration to an area of lower concentration, which does not require the input of energy. Concentration gradient, size of the particles that are diffusing, and temperature of the system affect the rate of diffusion.
How does concentration gradient affect the rate of active transport?
When active transport moves molecules and ions against their concentration gradient, a decreasing concentration gradient does not affect the rate of active transport. The rate of active transport is affected by: The speed of individual carrier proteins – the faster they work, the faster the rate of active transport.
How does concentration gradient affect the rate of diffusion in cells?
Extent of the concentration gradient: The greater the difference in concentration, the more rapid the diffusion. The closer the distribution of the material gets to equilibrium, the slower the rate of diffusion becomes. Lower temperatures decrease the energy of the molecules, thus decreasing the rate of diffusion.
How does concentration gradient affect the process of ATP synthesis?
The protons move with the concentration gradient, which allows them to do work- namely drive ATP synthesis. Imagine ATP synthase is like water wheel, as protons move by diffusion, the energy created allows them to do work. This process is called CHEMIOSMOSIS.
How are gradients important in diffusion and osmosis?
How are gradients important in diffusion and osmosis? Gradients allow materials to move across membranes, from an area of high concentration to an area of low concentration. Water will move out of a plant cell if it has a higher water potential (concentration) than the surrounding environment.
How does concentration gradient affect osmosis?
Concentration gradient – The movement of osmosis is affected by the concentration gradient; the lower the concentration of the solute within a solvent, the faster osmosis will occur in that solvent. Light and dark – They are also factors of osmosis; since the brighter the light, the faster osmosis takes place.
Does the rate of diffusion change as the concentration gradient changes?
Does the rate of diffusion change as the concentration gradient changes? Yes. An increase in the concentration gradient causes an increase in the diffusion rate. Smaller solutes diffuse faster than large ones for any given concentration gradient.
Does temperature affect the rate of diffusion?
When temperature increases, the kinetic energy of the particles has increased. The increased motion of the particles causes them to diffuse faster. Therefore, at higher temperatures, the rate at which fluid particles will diffuse is faster than at lower temperatures.
Which of these does not affect diffusion rate?
The factor that does not affect the rate of diffusion are the electrical charges of the diffusion particles.
What are the 4 factors that affect the rate of diffusion?
The rate of diffusion is affected by the concentration gradient, membrane permeability, temperature, and pressure. Diffusion takes place as long as there is a difference between the concentrations of a substance across a barrier.
Is active transport against the concentration gradient?
During active transport, substances move against the concentration gradient, from an area of low concentration to an area of high concentration. This process is “active” because it requires the use of energy (usually in the form of ATP). It is the opposite of passive transport.
Does temperature affect active transport?
Ion fluxes show a characteristically biochemical dependence on temperature when observed at the membrane level and over short periods after a perturbation of temperature. The primary active transport systems are enzymic and are dependent both on substrate supply and on changes in protein conformation.
How is active transport different from diffusion?
The main difference between diffusion and active transport is that diffusion is a passive transport method in which molecules move across the cell membrane through a concentration gradient whereas active transport requires cellular energy in order to transport molecules against the concentration gradient.
How does tonicity affect the function of the cell?
The tonicity of a solution is related to its effect on the volume of a cell. Solutions that do not change the volume of a cell are said to be isotonic. A hypotonic solution causes a cell to swell, whereas a hypertonic solution causes a cell to shrink.
What is the role of a concentration gradient?
The concentration gradient is an important process for understanding how particles and ions move in random motion in a solution or gas. It is the process used for particles moving from an area of higher concentration in a solution to an area of lower concentration.
How does concentration gradient represent potential energy?
The concentration gradient of a substance across a membrane represents potential energy because it drives diffusion.



