How do you find the valency of zinc?
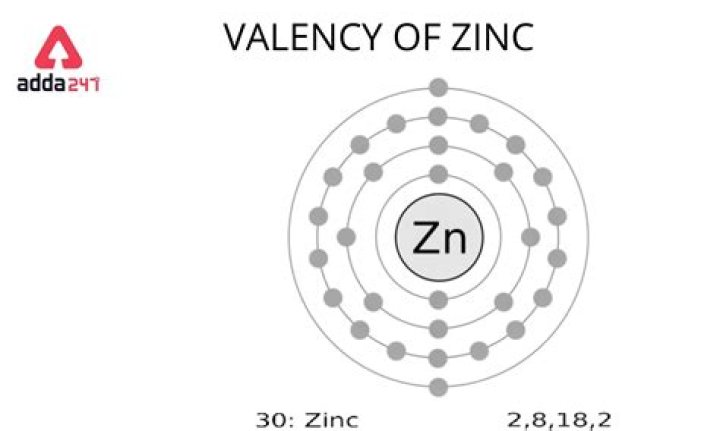
Zinc has 30 electrons I.e. 28 in first three shells and 2 in the fourth. To achieve octet, it must either lose two electrons or gain six. Since it is easier to lose two electrons, Zinc loses these two electrons in the outermost shell and forms a 2+ action. Hence, valency of Zinc is two (2).
Why valency of zinc is 2?
Answer: Zinc is a d-Block element and belongs to Transition Metal. Valence shell contains 2 electrons that is 4s2 that means that zinc can lose the two electrons located in the 4s-orbital to be become the Zn2+ cation. So, the valency of Zinc is 2.
What is the valency of Zn in zno?
The valence of zinc in zinc oxide is +2. Thus in another way it can be said thatZinc has the capacity to donate two electrons to oxygen and oxygen on gaining 2 electrons from it forms the compound zinc oxide.
Can zinc have a valency of 3?
Zinc traditionally has a valence of two, meaning two electrons take part in the element’s chemical reaction. A new article shows that the chemistry of zinc can be fundamentally changed, making it trivalent — or a valence of three — with the proper reagent.
Does zinc have variable valency?
Zinc does NOT show variable valency because of complete d subshell.
How many energy shells does zinc have?
Zinc atoms have 30 electrons and the shell structure is 2.8. 18.2. The ground state electron configuration of ground state gaseous neutral zinc is [Ar].
What is the valency of zinc and silver?
Valency of silver = 1. Valency of gold = 2. Valency of zinc = 2.
Does zinc have a full shell?
Zinc’s location reflects the fact that is has a completely filled third electron shell. Finishing your look at the electron configuration, you need to remember that the outer shell still only has two electrons even though the row of transition elements has filled in an additional ten electrons to the third shell.
How can we calculate valency?
Mathematically we can say that if the outermost shell of an atom contains 4 or less than 4 electrons, then the valency of an element is equal to the number of electrons present in the outermost shell and if it is greater than 4, then the valency of an element is determined by subtracting the total number of electrons
Which of the following is the reason for zinc not?
Answer: zinc atomic number is 30.It has configuration [Ar]4s2 3d10. If it loses 2es it has completely filled 3d shell and attain stability to exhibit more than oxygen state it should excite the electrons as the excitation energy is too high it does not exhibit variable oxygen state.
Does Cl show variable valency?
For chlorine, it has 7 electron in its outermost orbit. If it gains one electron, then it will acquire stable configuration like that of Argon. That is why, chlorine tends to gain one electron from other combining atoms, and hence its valency is 1. There are elements which can have varying valencies.



