fehling solution a and b formula, check these out | What is Fehling solution A and Fehling solution B?
Two solutions are required: Fehling’s “A” uses 7 g CuSO4. 5H2O dissolved in distilled water containing 2 drops of dilute sulfuric acid. Fehling’s “B” uses 35g of potassium tartrate and 12g of NaOH in 100 ml of distilled water.
What is Fehling solution A and Fehling solution B?
Fehling’s solution is prepared by combining two separate solutions: Fehling’s A, which is a deep blue aqueous solution of copper(II) sulfate, and Fehling’s B, which is a colorless solution of aqueous potassium sodium tartrate (also known as Rochelle salt) made strongly alkali with sodium hydroxide.
What is chemical formula of Fehling solution A and B?
It’s a mixture of two separate solutions: Solution A: aqueous solution of Copper (II) Sulphate (CuSO4) Solution B: aqueous potassium sodium tartrate KNaC4H4O6·4H2O (solution made alkaline by adding sodium hydroxide).
Why are Fehling’s A and B solutions made separately?
Fehling’s A and B solutions are kept separate because the bistartratocuprate (II) complex that is formed will easily degrade if they are combined. As it does not have a free Aldehyde or ketone group, Sucrose does not reduce Fehling’s solution.
What is the full name of Rochelle salt?
Rochelle salt, also called Sodium Potassium Tartrate Tetrahydrate, a crystalline solid having a large piezoelectric effect (electric charge induced on its surfaces by mechanical deformation due to pressure, twisting, or bending), making it useful in sensitive acoustical and vibrational devices.
What is Fehling’s solution Class 12?
So, Fehling solution is a mixture of copper sulphate, potassium sodium tartrate, and sodium hydroxide. It is used to differentiate between water-soluble carbohydrates (aldehyde) and ketone functional groups and as a test for reducing sugar.
How are ketones and aldehydes different?
You will remember that the difference between an aldehyde and a ketone is the presence of a hydrogen atom attached to the carbon-oxygen double bond in the aldehyde. Ketones don’t have that hydrogen. The presence of that hydrogen atom makes aldehydes very easy to oxidize (i.e., they are strong reducing agents).
What is Rochelle salt Class 12?
Sodium potassium tartrate is a double salt of tartaric acid with a chemical formula C4H4O6KNa·4H2O. It is also known as Rochelle salt or Potassium sodium tartrate. It is a colourless to white crystalline powder with a cool and saline taste. It has a pH value of 6.5 – 8.5.
What is Schiff reagent formula?
Fuschsine or rosaniline hydrochloride is a magenta-colored dye with a chemical formula C20H20N3·HCl, which is decolorized by sodium bisulfate [1,2].
How is Fehling factor calculated?
4.3.5 Calculations
Based on the factor for Fehling’s solution, total reducing sugars in V4 ml = 0.0025 × V1 g 21 Page 5 Practical Manual Food Chemistry and Physiology As 50 ml of the clarified and de-leaded solution is diluted twice (50 ml to 100 ml) after hydrolysis, dilution volume of the sample = (2 × V2).
What is the role of Fehling B solution in analysis of Aldehydic group?
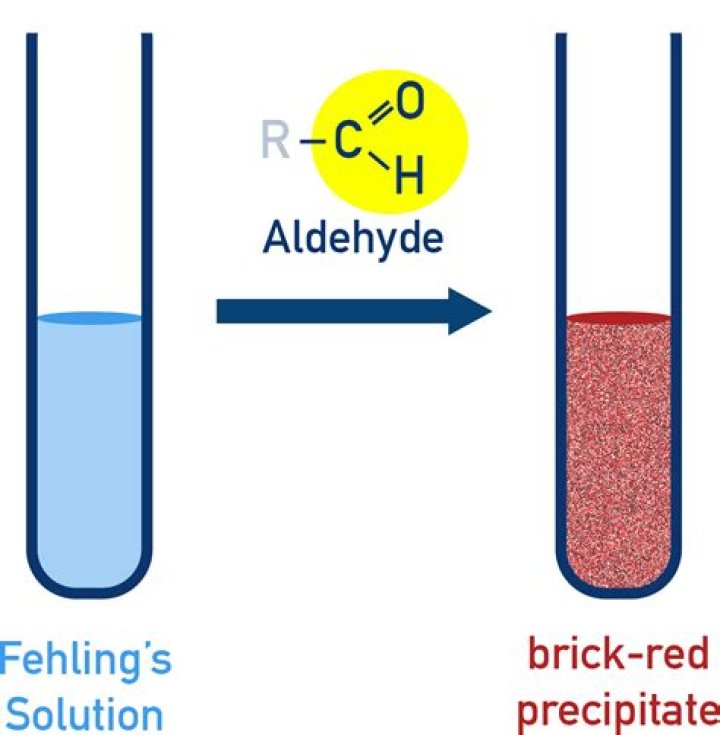
Fehling’s solution is a deep blue alkaline solution which is used to identify the presence of aldehydes or groups that contain any aldehyde functional group -CHO and in addition with Tollen’s reagent to differentiate between reducing and non-reducing sugars.
Why Fehling’s solution should be freshly prepared?
They are prepared whenever needed and not beforehand because this Fehling’s solution decomposes with time and becomes non-effective for the tests!
What is Fehling’s test and its purpose?
The Fehling test was developed in 1848 by Herrmann Feh- ling. It is a indicating reaction for reducing groups such as aldehyde functions. It makes it possible to differentiate be- tween reducing and non-reducing sugars.
Can you eat Rochelle salt?
A. Rochelle salt is potassium sodium tartrate. As an example of its assessed toxicity, it has an LD50 in rats (oral intake) of 1250 mg/kg, compared with salt (NaCl) of 3,000 mg/kg and sodium bicarbonate (often used as an indigestion treatment) of 4,420 mg/kg.
What is Rochelle salt tollens reagent?
Assertion (A): Rochelle’s salt is used as a complexing agent in Tollens reagent. Reason (R): Sodium potassium salt of tartaric acid is known as Rochelle’s salt. The IUPAC name of OH Rochelle’s salt N COOK is OC OH sodium potassium-2,3-dihydroxy butane-1,4-dioate.



