examples of neutralisation, check these out | What are some examples of neutralization?
Examples of Neutralization
Antacids.Treating Wasp Sting.Soil Treatment.Industrial Waste Treatment.Brushing Teeth.Shampoo and Conditioner.
What are some examples of neutralization?
19 Neutralization Reactions
acid + base → water + salt. HCl(aq) + KOH(aq) → H 2O(ℓ) + KCl(aq) 2 HCl(aq) + Mg(OH) 2(aq) → 2 H 2O(ℓ) + MgCl 2(aq) 3 HCl(aq) + Fe(OH) 3(s) → 3 H 2O(ℓ) + FeCl 3(aq) HCl(aq) + NaOH(aq) → H 2O(ℓ) + NaCl(aq) H +(aq) + Cl −(aq) + Na +(aq) + OH −(aq) → H 2O(ℓ) + Na +(aq) + Cl −(aq)
What is neutralization with two examples?
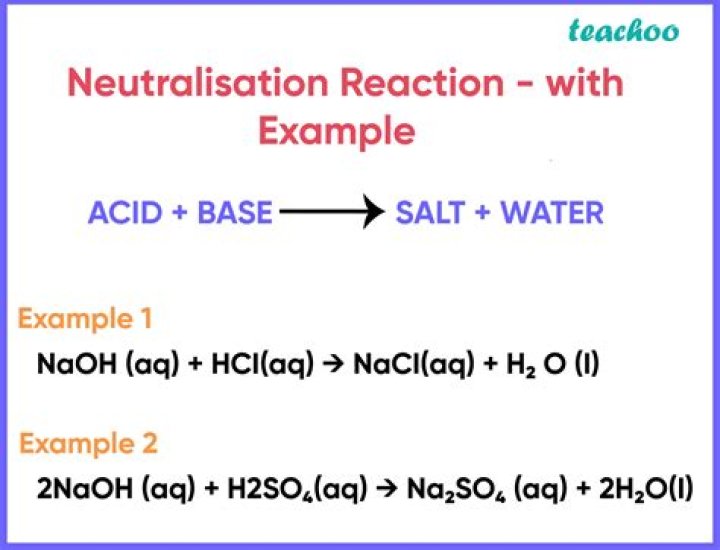
Since acid and base neutralize each other’s effect,it is called neutralization reaction. For Example: When Sodium Hydroxide ( NaOH) , a base, reacts with Hydrochloric acid (HCl) , it forms the salt, Sodium Chloride (NaCl) and Water.
What is one example of a neutralization reaction?
When a strong acid reacts with a strong base the resultant salt is neither acidic nor basic in nature i.e. it is neutral. For example when HCl (Hydrochloric acid), a strong acid, reacts with NaOH, a strong base, then the resulting salt is sodium chloride and water.
How is neutralisation used in everyday life?
Ans: The reaction between an acid and a base is called neutralisation. In everyday life, it is employed in different applications. For example, it is used in the neutralisation of stomach acidity, in the prevention of tooth decay, neutralising the soil, in the treatment of ant’s bite, etc.
What is Neutralisation one example Class 7?
The reaction between an acid and bases is called a neutralization reaction. The result of the reaction is salt and water. For example, when hydrochloric acid(acid) and sodium hydroxide(base) react they form sodium chloride(salt) and water. Also, heat is evolved.
Is HCl H2O a neutralization reaction?
Explanation: The reaction between sodium hydroxide (NaOH) and hydrochloric acid (HCl) is a neutralization reaction which results in the formation of a salt, sodium chloride (NaCl) , and water (H2O) .
What are some real life examples of acid base reactions?
Acid-Base Reactions
Whenever you combine an acid (e.g., vinegar, lemon juice, sulfuric acid, or muriatic acid) with a base (e.g., baking soda, soap, ammonia, or acetone), you are performing an acid-base reaction. These reactions neutralize the acid and base to yield salt and water.
What are salts give examples Class 7?
Ans: Salts are the ionic compounds generally formed by neutralisation of an acid with base. They can be acidic, basic as well as neutral. Example acidic salts: sodium bicarbonate, basic salts: magnesium chloride, neutral salt: sodium chloride.
What is phenolphthalein class 7th?
(c) Phenolphthalein: Phenolphthalein is an indicator solution which turns colourless in the presence of acids. Therefore, in the presence of sulphuric acid, phenolphthalein turns colourless.



