Does higher temperature mean lower viscosity?
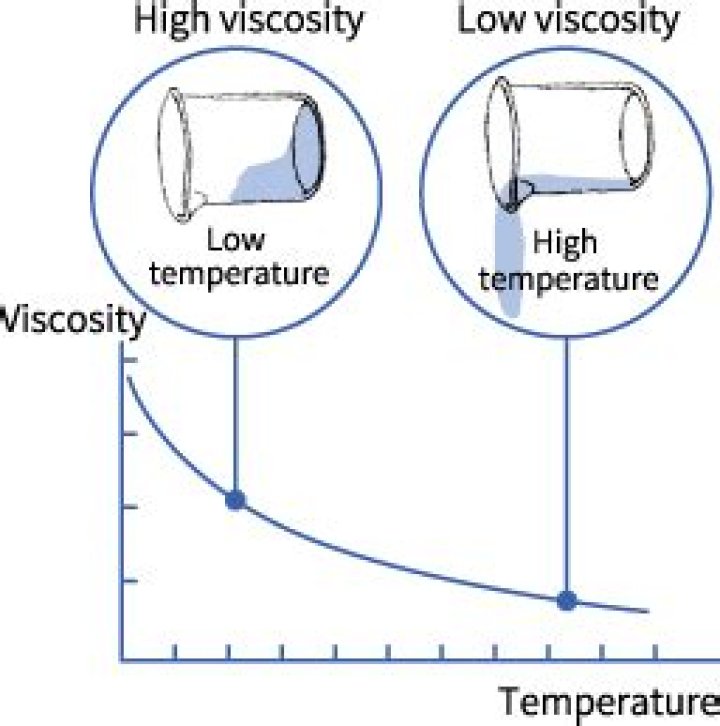
The liquid viscosity is highly affected by the heat. The viscosity decreases with an increase in temperature.
Does higher temperature mean higher viscosity?
With an increase in temperature, there is typically an increase in the molecular interchange as molecules move faster in higher temperatures. The gas viscosity will increase with temperature. With high temperatures, viscosity increases in gases and decreases in liquids, the drag force will do the same.
Why does viscosity decrease with temperature?
When temperature increases the energy level of liquid molecules increases and the distance between the molecule increases. It causes a decrease in intermolecular attraction between them, which reduce viscosity.
Viscosity and Temperature
For example, when syrup is cold it has a high viscosity and can be difficult to pour. When heated in a microwave, the viscosity decreases and the syrup flows more freely.
Does temperature affect viscosity?
The liquid viscosity is highly affected by the heat. The viscosity decreases with an increase in temperature.
The viscosity of liquids decreases rapidly with an increase in temperature, and the viscosity of gases increases with an increase in temperature. Thus, upon heating, liquids flow more easily, whereas gases flow more sluggishly.
When the temperature increases the viscosity of liquid?
When a liquid is heated, the kinetic energy of its molecules increases and the intermolecular attraction becomes weaker. Hence, the viscosity of a liquid decreases with increase in its temperature.
How does temperature affect viscosity of magma?
MAGMA VISCOSITY, TEMPERATURE, AND GAS CONTENT. The effect of temperature on viscosity is intuitive. Like most liquids, the higher the temperature, the more fluid a substance becomes, thus lowering its viscosity. Composition plays an even greater role in determining a magma’s viscosity.
In general, liquids tend to get “thinner” when their temperature increases. For example, honey and oil tend to flow better at higher temperatures. Therefore, increasing temperature decreases viscosity. Therefore, increasing temperature decreases density.
How does temperature affect the viscosity of oil?
Viscosity is a measure of an oil’s resistance to flow. It decreases (thins) with increasing temperature and increases (or thickens) with decreased temperature. The higher the VI of an oil, the less its viscosity is altered by temperature changes.
How does decrease in temperature of a liquid affects its viscosity?
In general, the viscosity of a simple liquid decreases with increasing temperature. Thus, as temperature increases, the average intermolecular forces decrease.
How does temperature affect cohesion?
Surface tension tends to reduce with increasing temperature. As a liquid heats up, the molecules in it speed up, which tends to break the bonds produced by cohesive forces.
Yes, water viscosity changes with temperature.
Water tends to have higher viscosity at lower temperatures and a lower viscosity at higher temperatures. Think of placing water in a freezer.
How does concentration affect viscosity?
The viscosity value at a given concentration follows H > A > G > E. An increase in solute concentration would result in an increase in viscosity, merely due to additional energy requirements to translate or rotate these molecules in solution.
How do you find the viscosity of a temperature?
Viscosity of water
Water has a viscosity of 0.0091 poise at 25 °C, or 1 centipoise at 20 °C. As a function of temperature T (K): (Pa·s) = A × 10B/(T−C) where A=2.414 × 10−5 Pa·s ; B = 247.8 K ; and C = 140 K .
What is effect of temperature?
The rate of a chemical reaction can be changed by altering the temperature. If the temperature is increased: the reactant particles move more quickly. they have more energy.
The viscosity of liquid decreases rapidly with rise of temperature.
When the temperature increased the surface tension of water?
– As we increase the temperature of the liquid, the molecules have more kinetic energy. That means that they have more velocity. This decreases the attraction between the molecules of the liquid. As a result there is a net decrease in the surface tension of water as we increase its temperature.



