Does glucose have a covalent bond?
The carbon atoms in the glucose ring each have four covalent bonds. The best, or optimum angle, between all these bonds is 109.5o, which results in a perfect tetrahedron. If, for any reason, these bonds are forced into greater, or smaller angles then the molecule will be strained or stressed, and be much less stable.
Is glucose a covalent bond?
Ionic or covalent? Glucose is a covalent compound because glucose contains 3 non-metals: oxygen, carbon, and hydrogen.
What type of bond is glucose?
All are in a single chain. So all the bonds present in glucose is double bond and covalent bond. Double bond is present in the O atom of ketone and covalent bonds connects carbon to carbon, carbon to hydrogen and oxygen to hydrogen.
Yes, this compound is known as glucose (a sugar). a). This compound is made of C-H bonds, C-O bonds and O-H bonds. All of these form covalent bonds because they share electrons and the difference in electronegativity values aren’t great enough to form ionic bonds.
Does glucose have ionic and covalent bonds?
Sugar, on the other hand, is composed of carbon, oxygen, and hydrogen and has covalent bonds. This type of bond is called an ionic bond. Ionic bonds usually form between metals and non-metals.
The carbon atoms in the glucose ring each have four covalent bonds. It follows, therefore, that the glucose molecule will be at its most stable when all the carbon atoms can arrange themselves so that their bond angles are all close to 109.5o.
Does glucose have a double bond?
Sugar Molecules
A single dash ( – ) represents a single bond, and a double dash (=) represents a double bond. This molecule of the sugar glucose consists of 6 carbon atoms bonded together as a chain with additional atoms of oxygen and hydrogen.
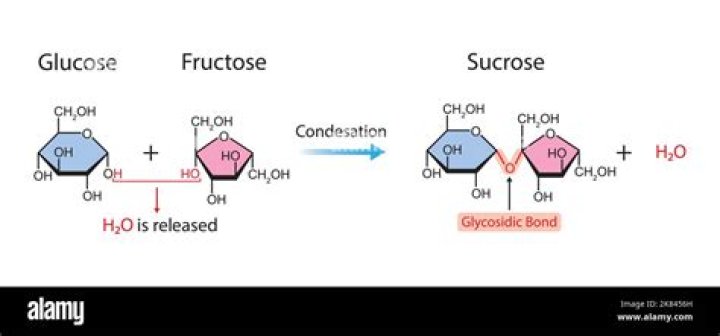
Does glucose have glycosidic bonds?
Most of the glucose units in glycogen are linked by α-1,4-glycosidic bonds. The branches are formed by α-1,6-glycosidic bonds, present about once in 10 units (Figure 11.13).
When glucose mole fraction is 0.003, all glucose molecules are hydrogen bonded with water molecules and there is no intermolecular hydrogen bonds between glucose molecules which is consistent with density profiles of glucose and water molecules in Fig.
Is fructose a covalent?
Glucose, galactose, and fructose are monosaccharide isomers, which means they all have the same chemical formula but differ structurally and chemically. Disaccharides form when two monosaccharides undergo a dehydration reaction (a condensation reaction); they are held together by a covalent bond.
Is so2 a covalent compound?
Sulfur dioxide is a Covalent compound because, in the case of sulfur dioxide, two atoms possessing similar electronegativity try to bond together. In doing so, the slight difference of electronegativity between the two atoms results in sharing of electron bonds forming covalent bonds.
Is glucose molecular?
Glucose is a simple sugar with the molecular formula C6H12O6. Glucose is the most abundant monosaccharide, a subcategory of carbohydrates.
By definition, an ionic bond is between a metal and a nonmetal, and a covalent bond is between 2 nonmetals. So you usually just look at the periodic table and determine whether your compound is made of a metal/nonmetal or is just 2 nonmetals.
Is glucose hydrophobic?
The major source of energy for mammalian cells is glucose. The transfer of glucose across the plasma membrane is necessary. Cell membrane is composed by lipid bilayer, which is hydrophobic. Glucose has hydrophilic nature.
What forms a nonpolar covalent bond?
A non-polar covalent bond is a type of chemical bond that is formed when electrons are shared equally between two atoms. Thus, in an atom, the number of electrons shared by the adjacent atoms will be the same. The covalent bond is also termed as nonpolar because the difference in electronegativity is mostly negligible.
What type of bond does fructose have?
In sucrose, the components glucose and fructose are linked via an acetal bond between C1 on the glucosyl subunit and C2 on the fructosyl unit. The bond is called a glycosidic linkage. Glucose exists predominantly as two isomeric “pyranoses” (α and β), but only one of these forms links to the fructose.



