define hess’s law, check these out | What is meant by Hess’s law?
What is meant by Hess’s law?
Hess’s law, also called Hess’s law of constant heat summation or Hess’s law of heat summation, rule first enunciated by Germain Henri Hess, a Swiss-born Russian chemist, in 1840, stating that the heat absorbed or evolved (or the change in enthalpy) in any chemical reaction is a fixed quantity and is independent of the
What is Hess law class 11?
The Hess’s law states that the total enthalpy change during a complete chemical reaction is the same regardless of the path taken by the chemical reaction.
What is Hess’s law give example?
As for example, the ethene gas and hydrogen chloride gas reacts to form chloroethane gas. The equation (ii) and (vii) are same, so the enthalpy change of formation of chloroethane gas from ethene gas and hydrogen chloride gas is -68.9 kJ/mol.
What is Hess law quizlet?
Hess’s Law. States that if reaction carried in series if steps, enthalpy change of overall reaction equals sum of enthalpy changes. Calculating enthalpy change for vast # reactions.
How do you Hess’s law?
Hess’s law states that if a process can be expressed as the sum of two or more steps, the enthalpy change for the overall process is the sum of the ΔH values for each step. To use Hess’s law, two principles must be understood: one, if an equation is reversed, the sign of the ΔH value is also reversed.
Which statement correctly describes Hess’s law?
Which option correctly describes Hess’s law? The total enthalpy change in a reaction is the sum of all of the enthalpy changes that occur in the intermediate steps of that reaction.
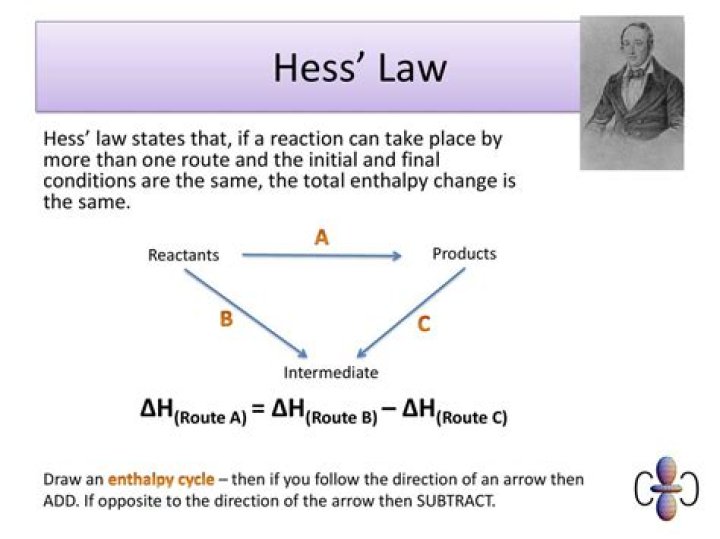
What is Hess law diagram?
Explaining Hess’s Law
Hess’s Law is saying that if you convert reactants A into products B, the overall enthalpy change will be exactly the same whether you do it in one step or two steps or however many steps. If you look at the change on an enthalpy diagram, that is actually fairly obvious.
What does Le Chatelier’s principle say?
Le Châtelier’s principle states that if a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium shifts to counteract the change to reestablish an equilibrium.
Is Hess law accurate?
Consistent with the law of energy conservation. If a procedure is written as the sum of many stepwise methods, the total process’s enthalpy change is equal to the sum of the different steps’ enthalpy changes. The law of Hess is true since enthalpy is a part of the state.
What is the main reason Hess’s law works?
Hess’s law is due to enthalpy being a state function, which allows us to calculate the overall change in enthalpy by simply summing up the changes for each step of the way, until product is formed. All steps have to proceed at the same temperature and the equations for the individual steps must balance out.
Which principle is Hess’s law based on?
Hess’s law states that energy changes are state functions. The amount of energy depends only on the states of the reactants and the state of the products, not on the intermediate steps. Energy (enthalpy) changes in chemical reactions are the same, regardless of whether the reactions occur in one or several steps.
How is Hess’s law applied for enthalpy?
Hess’s Law, also known as “Hess’s Law of Constant Heat Summation,” states that the total enthalpy of a chemical reaction is the sum of the enthalpy changes for the steps of the reaction. Therefore, you can find enthalpy change by breaking a reaction into component steps that have known enthalpy values.
How do you prove Hess’s law?
Hess’s law can be proved on the basis of first law of thermodynamics, according to which heat can neither be created nor destroyed. Let us suppose Q1 Joules is heat change when A changes to B directly and Q2 Joules is when A changes B indirectly via C.



