covalent modification, check these out | What is an example of covalent modification?
Covalent modifications are enzyme-catalysed alterations of synthesised proteins and include the addition or removal of chemical groups. Modifications can target a single type of amino acid or multiple amino acids and will change the chemical properties of the site.
What is an example of covalent modification?
The examples of the covalent modification strategy are acetylation/deacytilation; phosphorylation/dephosphorilation; myristoylation; ADP ribosylation; farnesylation; sulfation; ubiquitination. However, phosphorylation and acytilation are the most common examples.
What is covalent modification of DNA?
DNA methylation is a covalent chemical modification of DNA catalyzed by DNA methyltransferases (DNMTs). DNA methylation is associated with transcriptional silencing and has been studied extensively as a lifelong molecular information storage mechanism put in place during development.
What is the most common covalent modification?
Phosphorylation is a very common modification. In phosphorylation, a phosphate group is attached to an amino acid side chain. The most commonly phosphorylated side chain is a serine.
How enzyme activity is regulation by covalent modification?
Enzymes can be regulated by transfer of a molecule or atom from a donor to an amino acid side chain that serves as the acceptor of the transferred molecule. Another way of regulating an enzyme is by altering the amino acid sequence itself by proteolytic cleavage.
What is non-covalent modification?
There is another way for chemical modification: non-covalent modification with DNA-binding molecule. Instead of the covalently introduced functionalities, DNA is modified by non-covalent binding of small molecules bearing desired functionalities.
Is glycosylation a covalent modification?
It is a spontaneous reaction and a type of post-translational modification of proteins meaning it alters their structure and biological activity. It is the covalent attachment between the carbonil group of a reducing sugar (mainly glucose and fructose) and the amino acid side chain of the protein.
What is cytosine methylation?
Cytosine methylation is a common form of post-replicative DNA modification seen in both bacteria and eukaryotes. Modified cytosines have long been known to act as hotspots for mutations due to the high rate of spontaneous deamination of this base to thymine, resulting in a G/T mismatch.
What is DNA methylation?
DNA methylation refers to the addition of a methyl (CH3) group to the DNA strand itself, often to the fifth carbon atom of a cytosine ring. This conversion of cytosine bases to 5-methylcytosine is catalysed by DNA methyltransferases (DNMTs).
Is covalent modification allosteric?
Allosteric control:Allosteric enzymes contain distinct regulatory sites and multiple functional sites. Reversible Covalent Modification: The catalytic properties of enzymes can be altered by a covalent binding of a modifying group, most commonly to a phosphoryl group.
What does phosphorylation do to a protein?
Phosphorylation alters the structural confirmation of a protein, causing it to become activated ,deactivated or modifying it. Phosphorylation introduces a charged and hydrophilic group in the side chain of an amino acid, possibly changing a proteins structure by altering interactions with nearby amino acids.
How do you deactivate proteins?
Delete Content
The phosphorylation of a protein can make it active or inactive. Phosphorylation can either activate a protein (orange) or inactivate it (green). Kinase is an enzyme that phosphorylates proteins. Phosphatase is an enzyme that dephosphorylates proteins, effectively undoing the action of kinase.
What is proteolytic activation?
Proteolytic Activation is the activation of an enzyme by peptide cleavage. In this enzyme regulation process, the enzyme is shifted between the inactive and active state. Irreversible conversions can occur on inactive enzymes to become active. This inactive precursor is known as a zymogen or a proenzyme.
Which of the following is an example of regulating an enzyme by covalent modification?
Enzyme regulation by reversible covalent modification examples: Dinitrogen reductase: involved in nitrogen fixation in bacteria. Diptheria toxin: ADP-ribosylates and inactivates EF2.
Which enzyme is responsible for covalent modification of glycogen phosphorylase?
Regulation of glycogenolysis in skeletal muscle is dependent on a network of interacting enzymes and effectors that determine the relative activity of the enzyme phosphorylase. That enzyme is activated by phosphorylase kinase and inactivated by protein phosphatase-1 in a cyclic process of covalent modification.
Is proteolytic cleavage a covalent modification?
The blood clotting cascade and digestive enzymes (i.e chymotrypsin) work based on proteolytic cleavage. The other method of control is called reversible covalent modification. This involves the addition or removal of some type of group, most commonly the phosphoryl group, onto or from the enzyme.
What are the 3 main types of non-covalent interactions?
Non-covalent interactions can be classified into different categories, such as electrostatic, π-effects, van der Waals forces, and hydrophobic effects.
What is the difference between covalent and noncovalent bonds?
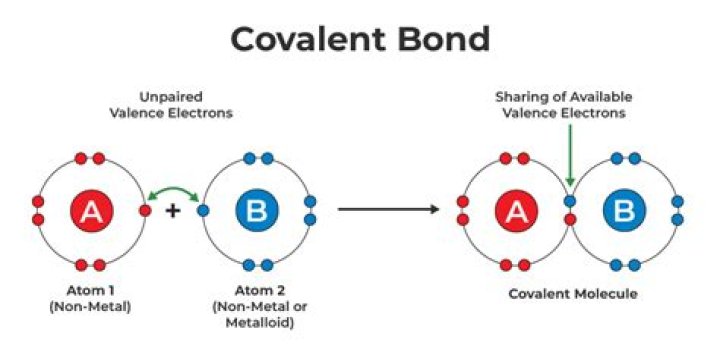
The key difference between covalent and noncovalent bonds is that covalent bonds form when two atoms share their electrons with each other whereas noncovalent bonds form either by completely exchanging electrons between two atoms or by not exchanging any electron.
Is non-covalent the same as ionic?
Noncovalent interactions arise via a number of different mechanisms. They include van der Walls interactions, hydrogen bonding, and electrostatic interactions (also called ionic bonding).



