br2 ccl4 reaction, check these out | What does Br2 and CCl4 do in a reaction?
What does Br2 and CCl4 do in a reaction?
Water contributes to bromohydrin formation, which further helps to add Br and OH to the double bond. As a result, the double bond breaks and the bromine atom binds to each carbon atom. The dark brown colour of bromine becomes colourless during this reaction.
What reacts with Br2 in CCl4?
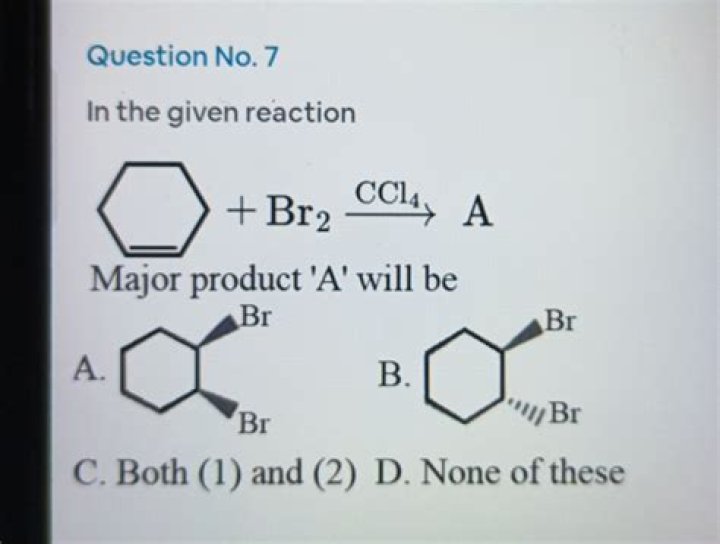
Description: Treatment of alkenes with bromine (Br2) gives vicinal dibromides (1,2-dibromides). Notes: The bromines add to opposite faces of the double bond (“anti addition”). Sometimes the solvent is mentioned in this reaction – a common solvent is carbon tetrachloride (CCl4).
What does Br2 and CCl4 do to alkene?
What do br2 and CCl4 do in a reaction? When Br2 interacts with CCl4, there is a dipole induced dipole interaction between the molecules and bromine gives Br + and Br, Br + attacks the alkene as an electrophile and undergoes an electrophilic addition reaction.
Is Br2 CCl4 syn addition?
Halogenation of alkene using Br2/ CCl4 is syn addition but not anti addition.
Would Br2 dissolve in CCl4?
Complete answer:Bromine dissolves in carbon tetrachloride, as both bromine and carbon tetrachloride are non-polar. Moreover, like dissolves in like, hence bromine dissolves in carbon tetrachloride.
What does Br2 FeBr3 do?
The bromine molecule reacts with FeBr3 by donating a pair of its electrons to the Lewis acid, which creates a more polar Br-Br bond, and thus a more reactive electrophile. Benzene will now attack this electrophile to generate the sigma complex.
Which does not give test of unsaturation with Br2 in CCl4?
Benzene does not decolorize a solution of bromine in CCl4. Because addition of bromine to benzene is endothermic reaction due to the loss of aromatic stability. Therefore, benzene does not undergo addition reaction with Br2. While addition of bromine to alkene is exothermic reaction at room temperature.
When CIS but 2 ene is treated with Br2 in CCl4 medium the product formed will be?
-The product formed is an enantiomeric pair as they are non-superimposable mirror images of each other. The pair is (2R, 3R) and (2S, 3S) dibromobutane. Hence the correct answer is: d) mixture of (2R, 3R) and (2S, 3S) dibromobutane.
How does Br2 react with alkenes?
Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The double bond breaks, and a bromine atom becomes attached to each carbon. The bromine loses its original red-brown color to give a colorless liquid.
Why do you think the reaction of an alkene with Br2 is called an addition reaction?
Explanation: Ethene and bromine are an addition reaction because ethene is an alkene – it has a double bond. It is easier for new atoms to open the double bond and react there than to remove the hydrogen already attached, and then bond to it, which would be a substitution reaction.
What happens when anthracene is treated with bromine CCl4?
all the hydrogen gets replaced of anthracene by br.
Which alkene will give meso form with Br2 CCl4?
trans-2-butene.
What is a syn addition?
Syn addition: An addition reaction in which all new bonds are formed on the same face of the reactant molecule.
What happens when HBr is added to 1 butene?
The reaction of 1 – butene with HBr in presence peroxides yields 1 – bromobutane.
Is Br2 soluble in alcohol?
Bromine is soluble in nonpolar solvents and in certain polar solvents such as alcohol and sulfuric acid.
Does NaCl dissolve in CCl4?
Since NaCl is polar in aqeous solution and Water is also polar so both dissolve each other. But CCl4 posses zero dipole moment hence non polar so NaCl is insoluble in it.
Which compound is more soluble in CCl4?
So, polar solutes are more soluble in polar solvents, while non-polar solutes are more soluble in non-polar solvents. So, looking at your choice of compounds, the non-polar substance will be most soluble in CCl4. That would be the hydrocarbon. All of the other compounds are polar or ionic.



