body centred cubic structure, check these out | Which is an example of body Centred cube?
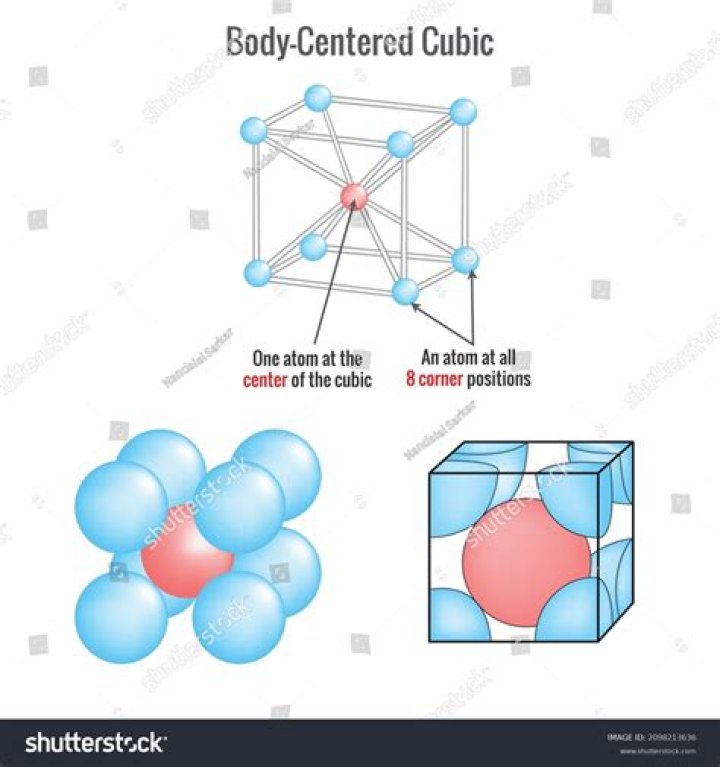
A body-centered cubic unit cell structure consists of atoms arranged in a cube where each corner of the cube shares an atom and with one atom positioned at the center. The atom at the corners of the cube are shared with eight other unit cells. As such, each corner atom represents one-eighth of an atom.
Which is an example of body Centred cube?
Sodium is an example of b.c.c. structure.
Which elements have body-centered cubic structure?
Examples of metals with the bcc structure are alpha iron, tungsten, chromium, and beta titanium.
How many atoms in body-centered cubic?
Body Centered Cubic
This unit cell uses nine atoms, eight of which are corner atoms (forming the cube) and one more in the center of the cube.
How do you know if a structure is FCC or bcc?
The BCC unit cell consists of a net total of two atoms, the one in the center and eight eighths from the corners. In the FCC arrangement, again there are eight atoms at corners of the unit cell and one atom centered in each of the faces. The atom in the face is shared with the adjacent cell.
Is gamma iron a body-centered cubic space lattice?
The alpha iron (α-Fe) is a body-centered cubic (BCC) and the gamma iron (γ-Fe) is a face-centered cubic (FCC).
What’s the packing efficiency of the body-centered cubic structure?
Packing efficiency of body centred cubic structure is $$68%.
What is the atomic radius of body-centered cubic structure?
The relation between edge length (a) and radius of atom (r) for BCC lattice is √3a=4r .
Is tungsten FCC or bcc?
In the solid state Tungsten has a body-centered cubic crystal structure with a = 0.316 nm and a nearest neighbor distance of 0.2.
Is bcc close-packed?
BCC structures have no close packed planes. Its coordination number is just the number of equidistant neighbors from the atom in the center of the unit cell.
Is CCP and fcc same?
Face Centered Cubic (fcc) or Cubic Close Packed (ccp) These are two different names for the same lattice. We can think of this cell as being made by inserting another atom into each face of the simple cubic lattice – hence the “face centered cubic” name.
What is the body-Centred cubic unit cell?
Body-centered cubic (BCC) is the name given to a type of atom arrangement found in nature. A body-centered cubic unit cell structure consists of atoms arranged in a cube where each corner of the cube shares an atom and with one atom positioned at the center.
Why is BCC more brittle than FCC?
BCC metals are generally more brittle. The main reason is their dislocation lattice friction stresses are higher than for FCC metals. Thus, dislocations move less readily in BCC lattices, which means less plasticity compared with FCC metals. BCC metals are generally more brittle.
Is steel a FCC or BCC?
The alpha phase is called ferrite. Ferrite is a common constituent in steels and has a Body Centred Cubic (BCC) structure [which is less densely packed than FCC]. Fe3C is called cementite and lastly (for us), the “eutectic like” mixture of alpha+cementite is called pearlite.
How many atoms do 6 unit cells of metal have in a body-centered cubic structure?
The body-centered cubic (bcc) has a coordination number of 8 and contains 2 atoms per unit cell. The simple cubic has a coordination number of 6 and contains 1 atom per unit cell.
Is ferrite an alpha iron?
Ferrite, also known as α-ferrite (α-Fe) or alpha iron, is a materials science term for pure iron, with a body-centered cubic B.C.C crystal structure. It is this crystalline structure which gives steel and cast iron their magnetic properties, and is the classic example of a ferromagnetic material.
What is the difference between alpha ferrite and delta ferrite?
This can begin within a temperature range of 900°C to 723°C, and alpha-ferrite is evident to room temperature. Delta ferrite is the high temperature form of iron, formed on cooling low carbon concentrations in iron-carbon alloys from the liquid state before transforming to austenite.
What type of space lattice is gamma iron?
the gamma iron (γ) is a face-centered cubic (FCC).



