bcl3 polar or nonpolar, check these out | Why BCl3 and BF3 are polar?
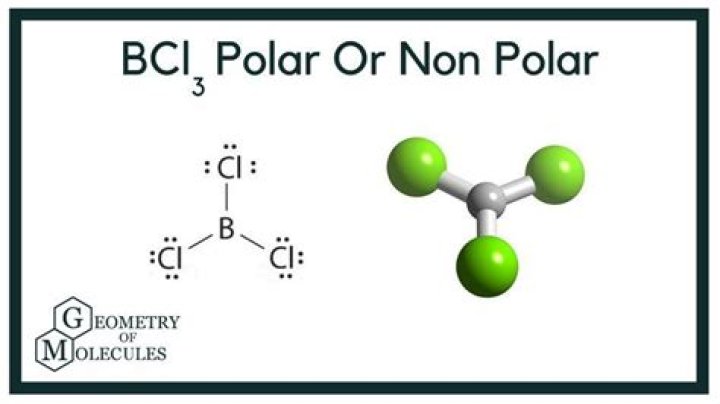
BCl3 is a nonpolar molecule. To have a polar bond, it needs to have an asymmetrical shapeshift in electron density to form an electrical dipole, but this is not the case for BCl3.
Why BCl3 and BF3 are polar?
So, is BCl3 polar or nonpolar? Boron Trichloride or BCl3 is a nonpolar compound because of its symmetrical structure ie; Trigonal Planar. The B-Cl bond itself is polar because of the difference in electronegativity of Boron(2.04) and Chlorine(3.16) atoms and all three B-Cl bonds lie at 120 degrees to each other.
Why BCl3 is nonpolar and PCl3 is polar?
The bond dipoles of three B-Cl bonds give a net sum of zero because the resultant of any two is equal and opposite to the third. Thus B-Cl3 does not have a dipole moment. On the other hand, in case of PCl3, the orbital dipole due to lone pair is in opposite direction to the resultant dipole moment of three P-Cl bonds.
Why is BCl3 nonpolar while ncl3 is polar?
The dipole moments of each of the Al-Cl bond is directed at 120 degree angles to each other in a plane, and is therefore cancelled out. Therefore it is a non-polar molecule.
Why are BF3 molecules non-polar?
BF3 is a non-polar compound. In BF3, the central boron atom has sp2 hybridised orbitals, resulting in an unfilled p orbital on the Bron atom and trigonal planar molecular geometry. Because the Boron-Fluorine bonds are all 120 degrees apart, any net dipole in that plane is cancelled out.
Is BrF3 polar or nonpolar?
Because of the existence of two lone pairs on the central bromine atom, BrF3 (bromine trifluoride) is a polar molecule with a deformed or twisted trigonal bipyramidal structure. And because the charge distribution on its atoms of BrF3 molecule is non-uniform, the BrF3 molecule is polar in nature.
Is ch4 polar or nonpolar?
All the outer atoms are the same – the same dipoles, and that the dipole moments are in the same direction – towards the carbon atom, the overall molecule becomes non-polar. Therefore, methane has non-polar bonds, and is non-polar overall.
Is BCl3 an ionic compound?
Boron and chlorine are both nonmetals, so this is a covalent compound.
Is BCl3 a zero dipole moment?
In BCl3, the central B atom undergoes sp2 hybridization which results in plane triangular geometry. The molecule has symmetry and the individual bond dipoles cancel each other. Hence, the molecule has zero dipole moment.
Does BCl3 have zero dipole moment?
Now BCl3 is a planar molecule in which the three B – Cl bonds are inclined at an angle of 120°. Therefore, the resultant of two B – Cl bonds in cancelled by equal and opposite dipole moment of the bond B – Cl bond as shown. Hence overall dipole moment of BCl3 is zero.
Is BCl3 or NCl3 more polar?
The BCl3 is a planar molecule whereas NCl3 is pyramidal becausea B-Cl bond is more polar than N-Cl bondb N-Cl bond is more covalent than B-Cl bondc nitrogen atom is smaller than boron atomsd BCl3 has no lone pair but NCl3 has a lone pair of electrons.
How many single bonds are in BCl3?
Thus we see that BCl3 contains three bonds, and there are no lone pairs of electrons on boron. The arrangement of three regions of high electron density gives a trigonal planar electron-pair geometry. The B–Cl bonds lie in a plane with 120° angles between them. BCl3 also has a trigonal planar molecular structure.
Why is BF3 nonpolar and PF3 is polar?
The BF3 molecule has a symmetrical trigonal planar geometry, like the SO3 molecule. In such a structure, the resultant moment of any two B-F dipoles is equal in magnitude but opposite in direction to the moment of the third one. So, the net dipole moment of the BF3 molecule is zero, and it is non-polar.
Why is BF3 nonpolar and PF3 polar?
So basically BF3 is non-polar because it has no lone pairs, its shape is symmetrical (triganol planar) and its dipoles cancel. PF3 is polar because it has one lone pair, it’s asymmetrical (pyramidal) and its dipoles don’t cancel?



