amphipathic cell membrane, check these out | Why is the cell membrane amphipathic?
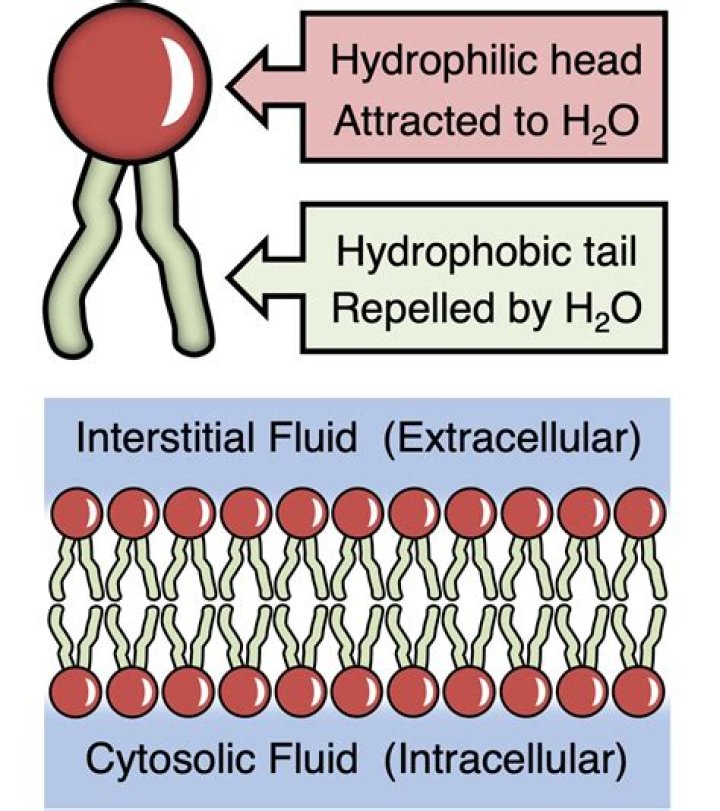
All of the lipid molecules in cell membranes are amphipathic (or amphiphilic)—that is, they have a hydrophilic (“water-loving”) or polar end and a hydrophobic (“water-fearing”) or nonpolar end. The most abundant membrane lipids are the phospholipids. These have a polar head group and two hydrophobic hydrocarbon tails.
Why is the cell membrane amphipathic?
The phosphate group on the phospholipid head is charged making it hydrophilic. This amphipathic nature allows for the bi- layer to form with the hydrophobic tails turning inwards away from the aqueous environment of the inside and outside of the cell with the hydrophilic phosphate head being in contact with the water.
What does amphipathic mean and how does it relate to the cell membrane?
Amphipathic is a word used to describe a chemical compound containing both polar (water-soluble) and nonpolar (not water-soluble) portions in its structure. It may also relate to a chemical compound having both hydrophobic and hydrophilic regions.
What is amphiphilic membrane?
Amphiphilic membrane is a kind of planar arrays of one or two monomolecular layers, which are derived from self-assembly of amphiphilic molecules, such as lipids, surfactants, or block copolymer amphiphiles, through noncovalent interactions in aqueous solution or in oil–water mixtures (Lipowsky et al.
What is an example of an amphipathic molecule?
Amphipathic or amphiphilic molecules have parts that are polar and nonpolar, making them both hydrophilic and lipophilic. Examples of amphipathic molecules include surfactants, phospholipids, and bile acids.
What do you mean by amphipathic?
/ (ˌæmfɪˈpæθɪk) / chem biochem of or relating to a molecule that possesses both hydrophobic and hydrophilic elements, such as are found in detergents, or phospholipids of biological membranes.
What is a phospholipid and why is it amphipathic?
A phospholipid is an amphipathic molecule which means it has both a hydrophobic and a hydrophilic component. The lipid tails, on the other hand, are uncharged, nonpolar, and hydrophobic, or “water fearing.” A hydrophobic molecule repels and is repelled by water.
Why is amphipathic important?
Amphipathic molecules are biologically useful because they can interact with both polar and non-polar substances. This allows them to make things possible that would not be possible with polar and non-polar molecules alone, including the creation of such crucial structures as the cell membrane.
What is the difference between amphoteric and amphipathic?
is that amphoteric is (chemistry) having the characteristics of both an acid and a base, and capable of reacting as either; amphiprotic while amphipathic is (chemistry) describing a molecule, such as a detergent, which has both hydrophobic and hydrophilic groups.
Why cholesterol is known as amphipathic molecules?
Cholesterol is referred to as an amphipathic molecule, that it contains its hydrophilic and hydrophobic parts. The hydroxyl group (-OH) in cholesterol is aligned with the phosphate head of the phospholipid on the cell membrane, which the rest of the cholesterol goes with the fatty acid of the membrane.
What is amphipathic molecules in chemistry?
Amphiphilic molecules is a general term that describes any compound that contains two distinct covalently bonded components with different affinity for the solvent in the same molecule, in which one part possesses a high affinity for polar solvents (such as water), and another part has a strong affinity for nonpolar
What does amphiphilic mean in chemistry?
Definition of amphiphilic
: of, relating to, or being a compound (such as a surfactant) consisting of molecules having a polar water-soluble group attached to a water-insoluble hydrocarbon chain also : being a molecule of such a compound.
What do amphiphiles do?
Amphiphiles are synthetic or natural molecules with the ability to self-assemble into a wide variety of structures including micelles, vesicles, nanotubes, nanofibers, and lamellae.
What are the 4 Biomacromolecules?
Examples of Biomacromolecules are Proteins, Nucleic Acids(DNA and RNA), Carbohydrates and lipids.
Where is amphipathic found?
Lesson Summary
Amphipathic molecules found in the body include lipids, which compose the cell membranes of cells, and cholesterol, which is necessary for hormone production and creates fluidity in the cell membrane.
What do amphipathic molecules possess?
All of the lipid molecules in cell membranes are amphipathic (or amphiphilic)—that is, they have a hydrophilic (“water-loving”) or polar end and a hydrophobic (“water-fearing”) or nonpolar end. The most abundant membrane lipids are the phospholipids. These have a polar head group and two hydrophobic hydrocarbon tails.
What is the purpose of integral proteins?
An integral protein, sometimes referred to as an integral membrane protein, is any protein which has a special functional region for the purpose of securing its position within the cellular membrane. In other words, an integral protein locks itself into the cellular membrane.
Which of the following is amphipathic?
Some of the examples of amphipathic molecules include bile salts, surfactants, and phospholipids. So, the correct answer is, ‘(c) Phospholipids’.



